Mini Review
Which Antimicrobial Peptides Have An Inhibitory Effect On Tuberculosis?
Bahar Sadegh Ehdaei1, Mehdi Shabani1 and Masoud Keikha2,3*
1Department of Microbiology and Virology, Isfahan University of Medical Sciences, Iran
2Antimicrobial Resistance Research Center, Mashhad University of Medical Sciences, Iran
3Department of Microbiology and Virology, Mashhad University of Medical Sciences, Iran
*Corresponding author: Masoud Keikha, Department of Microbiology and
Virology, Mashhad University of Medical Sciences, Mashhad, Iran, Tel:
09386836425; Email:
masoud.keykha90@gmail.com
Submitted: 19 April 2019; Accepted: 10 May 2019; Published: 13 May 2019
Cite this article: Ehdaei BS, Shabani M, xKeikha M (2019) Which Antimicrobial
Peptides Have An Inhibitory Effect On Tuberculosis? JSMC Biochem Mol Res 3: 4.
Tuberculosis (TB) remains as one of the important infectious
diseases which is considered as the ten top of cause of people dying
by WHO; According to WHO reports, there are approximately 10.7
million new TB cases in 2018, about two billion of people were
contaminated by Mycobacterium Tuberculosis which not clinical
symptoms but 5-10% of there are developed to reactivation of
tuberculosis [1,2]. In recent years, the advent and distribution of
drug-resistant TB (DR-TB) are converted to the major concern
of public health services throughout the world [2]. There are
558,000 Rifampin-resistant TB cases which 80% of these cases
have MDR-TB (Multi-drug resistant which is resistant to rifampin
and isoniazid) [1].
Antimicrobial peptides (AMPs) are oligopeptides which
expressed in eukaryotic cells particularly human and play a key
role as part of the innate immune response to infectious agents
[3]. This class of macromolecules has broad-spectrum antibiotics
effects which approved as novel therapeutic agents which
inhibits Gram-positive, Gram-negative, fungi and enveloped
virus [4]. According to the review of literature, AMPs have antimycobacterial
activities which could be efficient in susceptible
and MDR strains of M. tuberculosis [5]. In this report, we discussed about several antimicrobial peptides which approved
as a potent antibiotic for the treatment of tuberculosis.
The current tuberculosis treatment and anti-TB resistance
The conventional therapy of tuberculosis is consisting two
stages including the initial phase of rifampicin (RIF), isoniazid
(INH), pyrazinamide (PYZ), and ethambutol (ETB) which are
recommended daily for 2 months; followed preservative phase
of RIF and INH for additional 4 months [6]. Unfortunately, the
emergence and spread of Drug-resistant TB (DR-TB) particularly,
Multidrug-resistant TB (MDR-TB) which is resistant to both
isoniazid and rifampicin, and extensively drug-resistant TB (XDRTB)
that is MDR strains which are resistant to fluoroquinolone
and one of the second-line injectable are considered as a
major treat [7]. According to the review of literature, DR-TB is
commonly associated with high mortality rates; it estimated that
50% of MDR-TB cases and lower than 30% of patients with XDRTB
are survived [8]. MDR-TB and XDR-TB strains are mediated
by spontaneously point mutation in KatG (INH), rpoB (RIF), gyrB
(fluoroquinolones) and rrs (aminoglycosides) [9]. In addition,
side effect and limitation of anti-tuberculosis drugs have forced
to investing in a novel generation of ant-TB drugs; nowadays,
it is suggested that anti-microbial peptides are considered as
potential anti-tuberculosis drugs [10,11].
Defensins
Defensins are classified as Human neutrophil peptide (HNP)
and β-defencin; Human neutrophil peptide 1 (HNP-1) (peptide
sequence: ACYCRIPACIAGERRYGTCIYQGRLWAFCC) is a β-
sheeted cationic AMP which has antibacterial activity because
of three intramolecular disulfide bonds (Figure 1) [12]. Kalita et
al., have shown that MIC value of HNP1 against M. tuberculosis
H37Rv was 2.5 μg/ml which was higher than isoniazid (0.3 μg/
ml) and rifampicin (0.2 μg/ml) [13]. Also, there are four types of β-defencin (HBD1-4); Corrales-Garcia et al., were found
that HBD2 revealed sufficient bactericidal activity against M.
tuberculosis H37Rv (1.5 M) [14].
Cathelicidins
Cathelicidins are known as one of the great AMPs which was
purified form myeloid cell lines (bone marrow) for the first time;
LL-37(peptide sequence: LLGDFFRKSKEKIGKEFKRIVQRIKDFLRNLVPRTES)
peptide is derived from human cationic antimicrobial
protein which expressed by macrophages during entry of
pathogens which significantly reduces the intracellular survival of
M. tuberculosis [15]. Murine cathelicidin known as CRAMP (peptide
sequence: GLLRKGGEKIGEKLKKIGQKIKNFFQKLVPQPEQ)
has similar activity like to the human cathelicidin (Figure 1) [16].
Rivas-Santiago et al., have shown the bactericidal activity of LL-
37 and mouse CRAMP against a susceptible strain of Mtb H37Rv
and MDR strain during in vitro and in vivo studies [17].
Hepcidin
Hepcidin is a natural AMP (peptide sequence: DTHFPICIFCCGCCHRSKCGMCCKT)
compounds, which is expressed by the
liver cells during the inflammatory process (Figure 1) [18]. Sow
et al., have suggested that hepcidin could be reduced the CFUs of
MtbH37Rv with a concentration of 200 μg/ml [19].
-
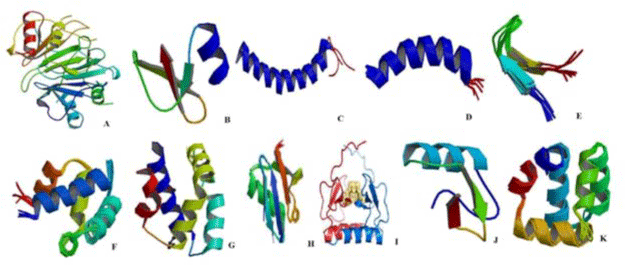 Figure 1: AMPs have ant-tuberculosis activity from A-L; A: Human neutrophil peptide, B: β-defencin, C: LL-37, D: CRAMP, E: Hepcidin, F: NK-lysin, G:
granulysin, H: Ubiquitin 2, I: CXCL1, J: Plectasin, K: AS-48; respectively. View Figure
Figure 1: AMPs have ant-tuberculosis activity from A-L; A: Human neutrophil peptide, B: β-defencin, C: LL-37, D: CRAMP, E: Hepcidin, F: NK-lysin, G:
granulysin, H: Ubiquitin 2, I: CXCL1, J: Plectasin, K: AS-48; respectively. View Figure
NK-lysin and granulysin
NK-lysin and granulysin are homologous cationic antimicrobial
peptides which are expressed by pig and human cytotoxic
lymphocytes respectively (Figure 1) [20]. NK-lysin (peptide sequence:
GYFCESCRKIIQKLEDMVGPQPNEDTVTQAASQVCDKLKILRGLCKKIMRSFLRRISWILTGKKAICVDIKICKE)
has destroyed
Mtb H37Rv during 8 days at 30 μM [21]. Granulysin (peptide sequence:
GRDYRTSLTIVQKLKKMVDKPTQRSVSNAATRVSR) has
cytolytic effects against both drug-susceptible M. tuberculosis
and MDR-TB [22]. Also, it suggested that Granulysin has a candidate
as a biomarker for diagnosis of TB [23].
Ubiquitin
Ubiquitin is naturally stored in secretory chromaffin granules
and released in response to different pathogens during Pro-inflammatory
process from chromaffin cells (Figure 1) [24]. Kieffer
et al., identified Ub2 in lysosome which plays a key role in autophagy
for Mtb lysis in phagolysosome [25]. Also, studies were
showed that synthesized peptide Ub2 (peptide sequence: STLHLVLRLRGG)
were antibacterial activity against tubercle bacillus [21].
There are numerous class of AMPs which show bactericidal
activity against both drug-susceptible strains and MDR-TB
strains which classified in different class including 1) Animal
AMPs (PR-39), 2) Human Immune cell-derived (LL37, mCRAMP,
E2, E6, Human neutrophil peptide 1 (HNP-1), Human Β-defensin
(hBD2), Granulysin, granF2, G13, Lactoferrin, IDR) Human Non
immune cell-derived (Ub2 (ubiquitndrived), Hepcidin), 4) Human
host defense (RNase 3, RNase 7, HCL2) , 5) Microbial AMPs
(Lacticin 3147, E50-52, Lassomycin, Bacteriocins (Bcn1-Bcn5), AS-48, NZX), 6) Synthetic AMPs (LLKKK-18, (LLKK)2, D5 (Synthetic
26-residue, amphipathic α-helical peptide), D-LAK analogs,
ATRA-1A, IDR-1018), 7) Mycobacteriophage AMPs (PK34, CHAP,
PlyG), lacticin 3147, E50-52 and human host defense ribonucleases
(RNase) [21,26-28].
In recent years, these studies are continuously performed and
noel AMPs are introduced with anti-mycobacterial effects against
TB such as CXCL1/CXCL2, NZX, and AS-48 which are novel potential
option for treatment of tuberculosis (Figure 1) [29-31].
CXCL1 / CXCL2
Boro et al., showed that chemokines of CXCL1 and CXCL2
were expressed during Mtb infection and recruit neutrophils to
the site of tubercle bacillus [29].
NZX as a novel derivative of Plectasin NZX (peptide sequence:
GFGCNGPWSEDDLRCHRHCKSIKGYRGGYCAKGGFVCKCY) is derivate
from Plectasin in fungi which has bactericidal effects on
M. tuberculosis both in vitro and in murine model studies [30].
Tenland et al., was found that the plectasin derivatives NZ2114
and NZX have antibacterial activities against Mtb (MIC concentrations
were 6.3 μM for H37Rv, 6.3 μM and 3.2 μM for two clinical
M. tuberculosis isolates and 6.3 μM for the clinical MDR isolate,
respectively); they found that NZX was less toxic to human cells
than LL37 [32].
AS-48
AS-48 is 70-aminoacid, an alpha-helical membrane-interacting
peptide which is extracted from Enterococcus faecalis and is
active against Gram-positive bacteria [31]. Aguilar-Pérez et al.,
have shown that the combination of AS-48 plus either lysozyme
or ethambutol has anti-TB activities [33].
During present decades it is suggested that numerous cyclic
peptides have antimycobacterial activities, for example,
griselmycins, depsidomycin, hytramycin, brunsvicamides, pyridomycin,
hirsutellide A and wollamides particularly, Wollamide B
which is evaluated in several studies [34].
Wollamide B
Wollamide B is cationic cyclohexapeptide which is extracted
from Streptomyces nov. strain MST-115088 by Khalil et al., in
2014 [35]. This compound has two uncommon residues such as
D-Orn, D-Leu or four L-amino acids in the structure [35-36]. For
the first time, Khalil et al., have shown that Wollamide B had IC50
of 3.1 μM against M. bovis. In recent years, Asfaw et al., suggested
that this compound has ani-TB effects during in vitro studies
(MICs ≤ 3.1 μM against Mtb H37 Rv) [34,35].
The cathelicidin-like antimicrobial protein of D. melanogaster (dCAMP)
Jin et al., was reported the cathelicidin-like antimicrobial protein
which is induced by Drosophila cg6568 gene during infection
with M. marinum infection in D. melanogaster; this compound is
cathelicidin like the activity that inhibits intracellular growth of
mycobacterial species [37].
Antimicrobial peptides are natural oligopeptides which have
the best candidate for the development of a novel natural generation
of antibiotics; According to the review of the literature, there
are 10 AMPs which entered clinical trials. In addition; there are
several AMPs with anti-tuberculosis activity (both of susceptible
and drug-resistant TB) provide the hypothesis that AMPs have
the best candidates for the treatment of tuberculosis.
 Figure 1: AMPs have ant-tuberculosis activity from A-L; A: Human neutrophil peptide, B: β-defencin, C: LL-37, D: CRAMP, E: Hepcidin, F: NK-lysin, G:
granulysin, H: Ubiquitin 2, I: CXCL1, J: Plectasin, K: AS-48; respectively. View Figure
Figure 1: AMPs have ant-tuberculosis activity from A-L; A: Human neutrophil peptide, B: β-defencin, C: LL-37, D: CRAMP, E: Hepcidin, F: NK-lysin, G:
granulysin, H: Ubiquitin 2, I: CXCL1, J: Plectasin, K: AS-48; respectively. View Figure