Review Article
Polyethylene glycol – Indocyanine green Nanoparticles for Photodynamic Therapy Technique
Mohammadreza Saboktakin*
Department of Nanomedicine, NanoBMat Company, Germany
*Corresponding author: Mohammadreza Saboktakin, NanoBMat
Company, GmbH, Hamburg, Germany, Email:
saboktakin123@gmail.com
Submitted: 13 May 2019; Accepted: 12 June 2019; Published: 15 June 2019
Cite this article: Saboktakin M (2019) Polyethylene glycol – Indocyanine green
Nanoparticles for Photodynamic Therapy Technique. JSMC Nanotechnol Nanomed 3: 5.
Nanoparticles (NPs) are synthetic materials with dimensions
from one to hundreds of nanometers, and remarkable
applications in biomedicine due to the unique way in which
they interact with matter [1,2]. There are currently more than
35 US FDA-approved NPs often incorporating polyethylene
glycol (PEG), with a larger number in preclinical studies for
both imaging and therapy (Figure 1A) [1,3-9]. NPs have large
payloads, stability, avidity, signal enhancement and the capacity
for multiple, simultaneous applications owing to their unique
size and high surface area: volume ratio [10]. While they are
bigger than molecules and many proteins, yet smaller than cells,
they behave differently to other therapies and imaging agents,
affecting their in vivo applications. For example, in cancer tissue,
NPs not only extravasate from the leaky tumor vasculature to a
higher degree than healthy tissue, but also remain in the area by
the enhanced permeability and retention (EPR) effect [11]. NPs
lodged in the tumor can then perform signaling and/or therapy
[10]. Despite these advantages, some fundamental challenges
hamper NP deployment to the clinic. These include uptake by
the reticuloendothelial system (RES), in which NPs are rapidly
shuttled out of circulation to the liver, spleen or bone marrow,
and nonspecific binding of NPs to nontargeted or nondiseased
areas. Concerns about NP toxicity often arise because of this RES
accumulation. Aggregation can lead to NP entrapment in the liver,
lungs or elsewhere due to capillary occlusion [12]. The addition of PEG to the NP surface (PEGylation) can reduce many of these
challenges (Figure 1B). PEG is a coiled polymer of repeating
ethylene ether units with dynamic conformations (Figure 1C).
In both drug-delivery and imaging applications, the addition of
PEG to NPs reduces RES uptake and increases circulation time
versus uncoated counterparts [13]. Aggregation decreases owing
to passivated surfaces, and association with nontargeted serum
and tissue proteins is diminished, resulting in so-called ‘stealth’
behavior. The PEG chains reduce the charge-based contact
typical of proteins and small-molecule interactions. Solubility in
buffer and serum increases due to the hydrophilic ethylene glycol
repeats and the EPR effect is modulated due to NP size changes
via addition of a PEG coat [14,15]. Due to these attributes,
PEGylated NPs generally accumulate in the liver a half to a third
of the amount of non- PEGylated NPs and demonstrate higher
tumor accumulation versus background [16]. PEG is inexpensive,
versatile and FDA approved for many applications [12]. All
NPs contain at least two fundamental spatial components:
the core and the corona that interact with the environment or
solvent. While core/shell, core/multishell systems add further
complexity, for example [18], all still possess an area in which
NP interfaces with the solvent (Figure 1B). PEG chains modify
this interface layer and increase circulation time. Circulation
half-time (t½) describes blood pool residence and is the period
over which the concentration of circulating NPs remains above
50% of the injected dose, analogous to a drug’s half-life [19]. NP
efficacy requires sufficient t½ to not only reach the target, but
also remain in the affected area (at concentrations sufficiently
above background tissue) long enough for image capture or drug
delivery. The RES system prevents site-specific accumulation
because it removes the NPs from circulation, acting as a
competitor to the intended target site [20]. In addition, the NPs
must clear from the nontargeted area to produce imaging contrast
or dosing efficiency. The ideal t½ is dependent on application. In
imaging, 2-6 h is optimal for injection, accumulation at targeted
site, clearance from nontargeted areas and data collection. The
ideal circulation time for therapeutic NPs is longer (days) to
allow repeated exposure to affected area. Unfortunately, this
can also expose healthy organ systems to the drug and is the
motivation for targeted NPs, as such systems preferentially accumulate in the diseased area. Approaches to measuring t½
vary with NP type. When labeled with radionuclides, g counting
of either specific organ systems or blood aliquots determines NP
circulation time. One limitation is dissociation of radionuclide
from NPs; however, radioactivity measurements may always be
carried out noninvasively [21].
Measurement of t½ via fluorescence, Raman, inductively
coupled plasma or chromatography/mass spectrometry is
very specific to the NP, but requires sequential sampling of the
blood pool. The RES is an immune system component, utilizing
circulating macrophages and monocytes, liver Kupffer cells and
spleen and other lymphatic vessels to remove foreign material,
such as bacteria and viruses, from the body [20]. Figure 2
illustrates how opsonin proteins associate with foreign bodies
and coat its surface [22]. As bacteria and viruses have the same
negative surface charge as phagocytic cells, opsonins are critical
to reducing the charge repulsion between the two systems [13].
Next, phagocytic cells engulf the material and transport it to the
liver or spleen for degradation and excretion (Figure 2 A3–A4).
Additional phagocytic macrophages are permanently located
in the liver. Known as Kupffer cells, these cells serve as a major
filter for many types of NPs and are a major interference with
long t½ [23]. The PEG polymer on a NP surface increases t½ by
reducing this opsonization process (Figure 2B2), thus preventing
recognition by monocytes and macrophages, allowing the NPs to
remain in the blood pool [13,22]. Hydrophobic particles are also
more vulnerable to the RES and hydrophilic PEG reduces these
complications [22]. In addition to NP-RES interactions, poor t½
can also result from NP-NP interactions (i.e., aggregation). NPs
aggregate primarily because the attraction between particles is
stronger than the attraction for solvent [13,24]. NPs with a high
surface energy have a greater tendency to aggregate as described
by the Derjaguin-Landau- Verwey-Overbeek (DLVO) theory
[25,26]. For spherical NPs, the interaction potential is related
to the electrostatic repulsive potential and the van der Waals
attraction potential [26]. PEG decreases the surface energy of
NPs and minimizes van der Waals attraction [27-29]. Aggregation can be induced by solvents of high (>100mM) ionic strength
(shielding of solvent from NP), highly concentrated solutions
of NPs (less distance between the NPs), time from synthesis, or
NP preparations with a very neutral (~±5 mV) zeta potential
[30]. PEG decreases the amount of attraction between NPs by
increasing the steric distance between them and increasing
hydrophilicity via ether repeats forming hydrogen bonds with
solvent. Other benefits to PEGylation include modifying the size
of the particle. The reduced renal filtration of particles larger
than 10 nm increases t½; however, at too large a size (>100 nm),
liver uptake increases and EPR extravasation may decrease [31].
PEG modifies the NP flexibility and the NP can become ‘softer’
after PEGylation than the underlying material, influencing
extravasation.
Prior to NP applications, PEG was used as a nontoxic, watersoluble
dispersant/stabilizer. Also known as Carboxwax®, it is
present in health and beauty aids, including laxatives, toothpastes
and eye drops, and is an excipient in tablet formulations [32].
PEG stabilizes organ and blood donations. Early work with
PEGylated NPs stemmed mostly from drug delivery [16,33-36].
One of the first reports on PEGylation was described by Davis and
Abuchowski [37,38], where they covalently attached methoxy-
PEGs (mPEGs) of 1900 and 5000 Da to bovine serum albumin and
to liver catalase. Later, acrylic microspheres functionalized with
PEG-modified human serum albumin increased t½ in vivo [39]. Li
and colleagues found that 75-nm latex particles remained in rat
circulation 40-times longer (half-life 20 min vs 13 h) when coated
than uncoated with PEG larger than 5000 kDa [33]. Klibanov and
Huang found that incorporation of dioleoyl N-(monomethoxy
polyethyleneglycol succinyl) phosphotidylethanolamine (PEGPE)
into posphatidylcholine: cholesterol liposomes (1:1) increased
t½ from 30 min to 5 h without increasing leakage of the liposome
interior [35]. In the mid- 1990s, Doxil® (liposomal delivery
vehicle for doxorubicin) and oncospar (PEG-l- asparaginase)
became the first FDA-approved NP therapeutics [40]. Doxil
increases doxorubicin bioavailability nearly 90-fold at 1 week from injection of PEGylated liposomes versus free drug [41]. The
use of PEG on the doxorubicin carrier yields a drug half-life of 72h
with circulation half-life of 36h [42,43]. Later, Abraxane® was
introduced as an albumin-functionalized NP for delivery of taxane
without cremphor to enhance drug efficiency [44]. Thadakapally
et al., developed a novel serum stable long circulating polymeric
nanoparticles for curcumin with a modification to the well known
and novel nanoparticle albumin bound technology. polyethylene
glycolalbumin- curcumin nanoparticles were prepared using
serum albumin and poly ethylene glycol using desolvation
technique. Nanoparticles were characterized for encapsulation
efficiency, particle size and surface morphology. Drug excipient
compatibility was determined using fourier transform infrared
spectroscopy. Physical state of the drug in the formulations was
known by differential scanning colorimetry. In vitro release
and solubility of the drug from nanoparticles were determined.
In vivo Drug release, tissue uptake and kupffer cell uptake was
determined with optimized nano formulation in rats after
intravenous administration. Cell viability assay was determined
using breast cancer cell line MD-MB-231. Entrapment efficiency
for prepared nanoparticle was above 95%. The polyethylene
glycol-albumin-curcumin nanoparticles exhibited an interesting
release profile with small initial burst followed by slower and
controlled release. Solubility of the drug from the formulation
was increased. A sustained release of drug from nanoparticles
was observed for 35 days in both in vitro and in vivo studies
with the optimized formulation. Polyethylene glycol-albumincurcumin
nanoparticles showed lesser liver and kupffer cell
uptake as compared to that of curcumin-albumin nanoparticles
suggesting the bestowment of stealthness to nanoparticles with
pegylation. Also, the antiproliferative activity of polyethylene
glycol-albumin- curcumin nanoparticle formulation was more
as compared to native curcumin. Polyethylene glycol-albumincurcumin
nanoparticles thus developed can be conveniently
used in breast cancer with improved efficacy compared to
conventional therapies and as an alternate to nanoparticle
albumin bound technology which is used in producing Abraxane,
albumin based breast cancer targeting nanoparticles of paclitaxel
[45]. Photothermal therapy (PTT) and photodynamic therapy (PDT) are emerging physical tumor treatments utilizing near
infrared (NIR) light-absorbing agents which lead to thermal
ablation of cancer cells or generate highly reactive oxygen
species (ROS) via photosensitizer to ablate tumors [46,47]. PTT
and PDT possess several advantages, such as minimal invasion,
high therapeutic efficacy, limited side-effects, selective localized
treatment and reproducible properties [48,49], and hence have
received much attention in recent years [50,51]. Until now, a
variety of materials has been explored as PTT or PDT agents due
to their high absorption in the tissue- transparent NIR wavelength
range, including organic fluorescent dyes [52], gold nanorods
[53], CuS nanoparticles (NPs) [54], polymer NPs [55], carbon
nanomaterials [56], etc. [57,58]. However, fluorescent dyes
may be removed rapidly from the systemic circulation and lack
specificity to a tumor, and inorganic photothermal agents have
potential long-term toxicity due to the difficulty of degrading in
the body [59]. Therefore, exploiting biocompatible and targeted
therapeutic nanoagents with enhanced photothermal conversion
capability and ROS generation ability to amplify PTT and PDT
treatments remains challenging. Indocyanine green (ICG) is a
clinical infrared imaging agent approved by the U.S. Food and Drug
Administration (FDA), and has been applied in optical imaging of
human vasculature, tissue and cells due to its biocompatibility
and unique optical properties [60]. Due to strong absorption at
780 nm, ICG can effectively convert absorbed NIR optical energy
into heat for PTT [61], and produce ROS for PDT [62], under
NIR laser irradiation [63]. Nevertheless, the application of ICG
in tumor phototherapy is limited by its tendency to aggregate,
rapid degradation in aqueous solution [64], poor photo-stability
and non-specific binding to proteins [65]. To overcome those
limitations, various nanoparticle delivery systems have been
developed to encapsulate ICG [66]. Lv et al., used a mesoporous
silica (mSiO2) matrix to load ICG molecules, and demonstrated
that loaded ICG displayed a more enhanced photothermal effect
than pure ICG [67]. ICG-loaded mesoporous silica NPs also could
not only limit the degradation of ICG, but reach and stay at a tumor
for a long period of time due to an enhanced permeability and
retention (EPR) effect [68]. Hence, loading of ICG within targeting nanocarriers with high efficiency is shown to be an effective way
to promote the application of ICG in PTT and PDT treatment.
To improve the efficacy of PTT, polyethylene glycol (PEG)
nanoparticles (NPs), indocyanine green (ICG) for NIR laserinduced
PTT. ICG after administered intravenously will be readily
bound with blood proteins and hence leads to only 2 ± 4 min of
plasmatic half-life. Among various pharmaceutical polymers,
PEG is one of the best defined biomaterials with FDA approval for
drug encapsulation due to its biocompatibility, biodegradability,
and controllability for drug release.
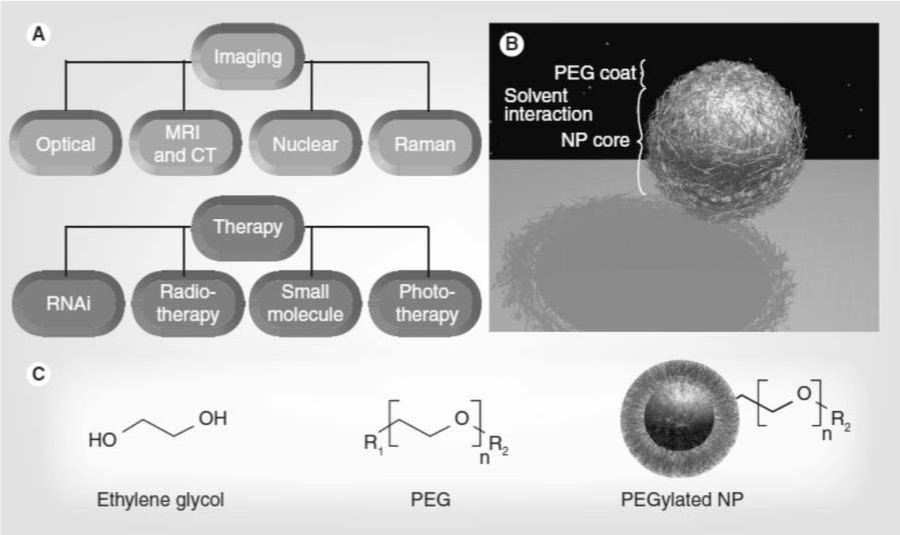 Figure 1: Nanoparticle applications of Polyethylene glycol. View Figure
Figure 1: Nanoparticle applications of Polyethylene glycol. View Figure
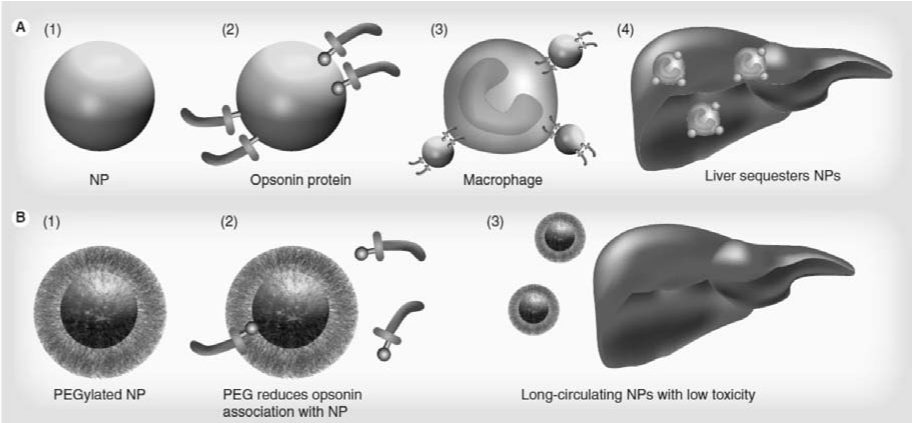 Figure 2: Polyethylene glycol prevents uptake by the reticuloedothelial system. View Figure
Figure 2: Polyethylene glycol prevents uptake by the reticuloedothelial system. View Figure