Among various approaches of breast cancer treatment, nearinfrared
(NIR)- mediated phototherapy is one of the most
promising strategies for serving as a supplement to traditional
cancer therapies since it can provide 1) enhanced tissue
penetration efficacy as compared with that operated by visible
light and 2) moderate toxicity to normal cells/tissues through
use of targeted photosensitive agents and/or spatially controlled
light irradiation [1]. Generally speaking, phototherapy is carried
out by hyperthermia and/or reactive oxygen species (ROS)
generated from the photosensitizers under light illumination in
the presence of oxygen that the former may cause thermal
ablation of cancer cells (i.e., photothermal therapy; PTT), while
the latter may seriously interfere cellular metabolism and thus
trigger programed cell death (i.e., photodynamic therapy; PDT)
[2,3]. No matter which mechanism is utilized, the photosensitizer
plays a key role in the effectiveness of phototherapy. Indocyanine
green (ICG) is an U.S. Food and Drug Administration (FDA)-
approved tricarbocyanine dye which enables to absorb and
fluoresce in the region of 650 ± 850nm. Currently, in addition to
serving as a fluorophoric agent for use in diagnostic purposes
such as NIR image-guided oncologic surgery [4], fluorescence
angiography [5], and lymph node detection of cancer [6], ICG has
been exploited as a photosensitizer for use in cancerous phototherapy including breast, brain, and skin tumors [7-9] since
it enables to produce heat and ROS (i.e., singlet oxygen) upon NIR
irradiation. Although ICG is of particular advantage for use in
cancer phototherapy, it adversely tends to disintegrate in
aqueous medium and such degradation can be markedly
accelerated by light irradiation (photodegradation) and/or
heating (thermal degradation) [10]. Furthermore, ICG after
administered intravenously will be readily bound with blood
proteins and hence leads to only 2 ± 4 min of plasmatic half-life
[11,12]. These circumstances seriously hinder the applicability of
ICG in the clinic and thus a strategy that enables to enhance the
aqueous stability and target efficiency of ICG is certainly needed
for ICG-mediated therapy. Nanomedicine may offer a feasible
means for usage of ICG without aforementioned defects since it
may provide merits of enhanced bioavailability, improved
stability, and security for the payload [13]. In terms of the
materials used for making drug carrier, polymer is often
considered as the preferred candidate since it can be manipulated
to tailor the properties and/or functionalities required by the
product [14]. Among various pharmaceutical polymers, poly
(lactic-co-glycolic acid) (PLGA) is the copolymer of poly(lactic
acid) and poly(glycolic acid) and is one of the best defined
biomaterials with FDA approval for drug encapsulation due to its
biocompatibility, biodegradability, and controllability for drug
release [15]. Polyethylene glycol (PEG), another FDA-approved
polymer with characteristics of nontoxicity and less
immunogenicity, is frequently used for surface modification of
drug carrier since the retention time of the PEG-coated particle in
the blood circulation can be markedly increased [16]. Taken all
together, we aim to develop an anti-HER2 ICG-encapsulated PEGcoated
PLGA nanoparticles (HIPPNPs) for targeted phototherapy
of HER2-expressing breast cancer cells. The use of ICG by
implantation of HIPPNPs instead of naked molecules is
advantageous because the polymeric carrier (i.e., HIPPNP) may
1) potentially protect the entrapped ICG from degradation caused
by external stimuli such as light, heat, and/or extreme pH [17]; 2)
preciously localize the therapeutic region to reduce off-target
toxicity, and 3) provide accurate estimation for the efficacy of
ICG-mediated phototherapy. There are many of reports that ICG encapsulators, such as (poly (lactic-co-glycolic acid) (PLGA)
nanoparticles (diameter ~360nm) and silica-polymer composite
microcapsules (diameter ~0.6 to 2μm) improve the molecular
instability of ICG and prolong its plasma half-life [18,19].
However, both of these nanoparticles are limited in size for in
vivo tumor imaging depending on their EPR effects. Recently,
several publications have reported promising results using
smaller nanoparticles to encapsulate ICG for in vivo imaging. For
example, Zheng et al. [20], developed ICG encapsulated PLGAlipid
nanoparticles conjugated with folic acid (FA) and
demonsstrated their use as NIR contrast agents for tumor
diagnosis and targeted imaging. Altinoglu et al., also synthesized
biodegradable calcium phosphosilicate nanoparticles (CPNPs)
and demonstrated that small size (16nm) ICG- encapsulating
CPNPs have significantly better contrast agent optical properties
than free fluorophores for tumor imaging [21]. Other inorganic
delivery systems using silica nanoparticles have been developed
to encapsulate ICG, and the ICG-SiO2 nanoparticles have the
potential to be used as contrast agents for optical NIR imaging as
well [22]. Among these nanocarriers, micelles are one of the
successful types of drug delivery systems for in vivo applications
due to their small size (approximately 10-100nm), which reduce
clearance by the reticuloendothelial system (RES) and allow for
an enhanced EPR effect [23,24]. Therefore, the encapsulation and
stabilization of ICG dye as a contrast agent in micellar systems is
of particular interest. For example, Pluronic F-127 (PF-127)
polymeric micelles are approved by the FDA and have been
successfully demonstrated to encapsulate and stabilize ICG as an
NIR contrast agent for optical imaging [25,26]. Encapsulation of
ICG within various micellar systems was also investigated by
Kirchherr and co-workers, and they found many micellar systems
improved the optical properties and stability of the ICG [27].
More interestingly, Zheng et al. have recently reported a dualfunctional
ICG-PL-PEG agent with several unique features for
optical imaging and photo-therapy [28]. This may emerge as a
new strategy for combining tumor treatment and diagnosis
together, using nanovectors with ICG. In summary, this editorial
discussed recent developments in nanocarrier ICG contrast
agents for NIR optical imaging. Here just some of the areas are
collected in terms of subjects and interests but it is hoped that
every reader will find something of interest to them. Manchanda
et al. [29], have recently reported the fabrication of a novel
polymer nanoparticle delivery system with simultaneously
entrapped indocyanine green (ICG) and doxorubicin (DOX). This
system has potential applications for combined chemotherapy
and hyperthermia. Research in our group showed that
simultaneous use of ICG and DOX with localized hyperthermia
can produce the same effect as that achieved by larger doses of
chemotherapy alone. The potential of dual- agent PLGA
nanoparticles (ICG-DOXPLGANPs) has explored to overcome
multidrug resistance (MDR) mechanisms in cancer cells by
increasing intracellular drug concentrations via nanoparticle
uptake. ICG-DOX-PLGANPs were prepared by the O/W emulsion
solvent evaporation method. The dominant processing
parameters that control particle size and drug entrapment
efficiencies of ICG and DOX were PLGA concentration, PVA
concentration and initial drug content. The previous formulation optimized based on those parameters. Entrapment efficiency of
the optimized ICG-DOX-PLGANPs was measured by fluorescence
measurements using the DMSO burst release procedure. The
internalization of ICG-DOX- PLGANPs by three cancer cell lines
was visualized by confocal laser microscopy and fluorescence
microscopy. Cytotoxicity was assessed using the SRB assay. The
nanoparticles produced by optimal formulation had sizes of 135
± 2 nm, (n=3) with a low poly-dispersity index (0.149 ± 0.014,
n=3) and a zeta potential of -11.67 ± 1.8 mV. Drug loading was
approximately 3% w/w for ICG and 4% w/w for DOX (n=3).
Cellular uptake of ICG and DOX from ICG-DOX-PLGANPs in DOXresistant
MESSA/Dx5 cancer cells was higher compared to free
ICG and free DOX treatment. However, the same phenomenon
was not observed in MES- SA and SKOV-3 cancer cell lines. The
SRB cytotoxicity results show that ICG- DOX-PLGANPs are more
toxic than free DOX in DOX-resistant cell lines. In the development
of drugs for intra-articular administration, sustained-release
formulations are desirable because it is difficult to maintain the
effect of conventional injections due to immediate drug leakage
from the joint cavity. In this study, a sustained-release poly
(lactic-co-glycolic acid) (PLGA) microsphere formulation for
intra-articular administration containing indocyanine green
(ICG) as a model drug was prepared to follow its fate after intraarticular
administration in rats with a real-time in-vivo imaging
system. ICG administered as an aqueous solution leaked from the
joint cavity in a short time and was excreted outside the body
within 1-3 d. However, ICG in the sustained-release formulation
was retained in the joint cavity and released for 2 weeks. Next, a
sustained-release formulation containing PLGA microspheres in
a hyaluronic acid (HA) gel formulation was prepared [30] (Figure 1).
Ronak et al. [31], have been developed and characterized
multifunctional biodegradable and biocompatible poly lacticco-
glycolic acid (PLGA) nanoparticles loaded with indocyanine
green (ICG) as an optical-imaging contrast agent for cancer
imaging and as a photothermal therapy agent for cancer
treatment. PLGA-ICG nanoparticles (PIN) were synthesized
with a particle diameter of 246 ± 11nm, a polydispersity index
of 0.10 ± 0.03, and ICG loading efficiency of 48.75 ± 5.48%. PIN
were optically characterized with peak excitation and emission at 765 and 810 ± 5 nm, a fluorescence lifetime of 0.30 ± 0.01 ns,
and peak absorbance at 780 nm. The cytocompatibility study of
PIN showed 85% cell viability till 1mg∕ml concentration of PIN.
Successful cellular uptake of ligand conjugated PIN by prostate
cancer cells (PC3) was also obtained. Both phantom- based and in
vitro cell culture results demonstrated that PIN (1) have the great
potential to induce local hyperthermia (i.e., temperature increase
of 8 to 10°C) in tissue within 5mm both in radius and in depth;
(2) result in improved optical stability, excellent biocompatibility
with healthy cells, and a great targeting capability; (3) have
the ability to serve as an image contrast agent for deep-tissue
imaging in diffuse optical tomography (Figure 2).
Nowadays, a new technique such as photodynamic therapy
(PDT) is used to achieve effective root canal disinfection
and eliminate Enterococcus faecalis as the most prevalent
species associated with secondary endodontic infections and
treatment failures. Employment of an optimized nontoxic
photosensitizer (PS) such as indocyanine green (ICG) is a crucial
part of this technique; the current study aimed at improving ICG
photodynamic properties through conjugation of ICG into nanographene
oxide (nGO) as a new PS, to evaluate the antimicrobial
effects of nGO/ICG against E. faecalis. The PDT based on ICG, at
the concentration loaded in nGO, showed a significant reduction
in the number of E. faecalis at a lower concentration of ICG. In
conclusion, the application of nGO as a new drug delivery system,
in addition to the anti-bacterial property, offers other benefits
such as cost beneficial outcomes due to using the lower dye
concentration (less toxicity), and less tooth discoloration. The
nGO/ICG-PDT could be proposed as a new approach to treat
endodontic infections, alone or in combination with conventional
root canal treatments [32] (Figure 3).
Zheng et al. [33], had successfully constructed the
fluorescence image guided photothermal therapy reagents
based on (WO+ICG)@PLGA) nanoparticles. In our design, to
improve their tumor targeting, the macrophages as cell-based
biocarriers were employed for delivery the (WO+ICG)@PLGA
nanoparticles. The macrophages carried these nanoparticles still
had phagocytosis to tumor cells and could also secret plenty of
anti-tumor cytokines for immunotherapy of carcinoma. They
also further elucidated the superior solid tumor suppression
efficiency of the (WO+ICG)@PLGA nanoparticles loaded macrophages targeting biocarriers delivery system in vivo. The
system achieved a significant antitumor effect by activating
immunotherapy and photothermal therapy in vivo. Hence, such
kind of (WO+ICG)@PLGA nanoparticles loaded macrophages
delivery system has great potential applications as targeting
biocarriers loading drugs and imaging agents for visual-guided
synergistic therapy in vivo (Figure 4).
The photothermal heating and release properties of
biocompatible organic nanoparticles, doped with a near
infrared croconaine (Croc) dye, were compared with analogous
nanoparticles doped with the common near-infrared dyes ICG
and IR780. Separate formulations of lipid−polymer hybrid
nanoparticles and liposomes, each containing Croc dye, absorbed
strongly at 808nm and generated clean laser-induced heating (no
production of 1O2 and no photobleaching of the dye). In contrast,
laser-induced heating of nanoparticles containing ICG or IR780
produced reactive 1O2, leading to bleaching of the dye and also
decomposition of coencapsulated payload such as the drug
doxorubicin. Croc dye was especially useful as a photothermal agent for laser-controlled release of chemically sensitive payload
from nanoparticles. Solution state experiments demonstrated
repetitive fractional release of water-soluble fluorescent dye
from the interior of thermosensitive liposomes. Additional
experiments used a focused laser beam to control leakage from
immobilized liposomes with very high spatial and temporal
precision. The results indicate that fractional photothermal
leakage from nanoparticles doped with Croc dye is a promising
method for a range of controlled release applications [34] (Figure 5).
Near-infrared (NIR) laser-induced photothermal therapy
(PTT) uses a photothermal agent to convert optical energy into
thermal energy and has great potential as an effective local,
minimally invasive treatment modality for killing cancer cells.
To improve the efficacy of PTT, Chengcheng Niu et al. [35],
developed poly (lactide co-glycolide) (PLGA) nanoparticles
(NPs) encapsulating super paramagnetic iron oxide (Fe3O4),
indocyanine green (ICG), and perfluoropentane (PFP) as
synergistic agents for NIR laser-induced PTT. They fabricated
a novel type of phase-shifting fluorescent magnetic NPs, Fe3O4/
ICG@PLGA/PFP NPs that effectively produce heat in response
to NIR laser irradiation for an enhanced thermal ablation effect
and a phase-shift thermoelastic expansion effect, and thus, can be
used as a photothermal agent (Figure 6).
-
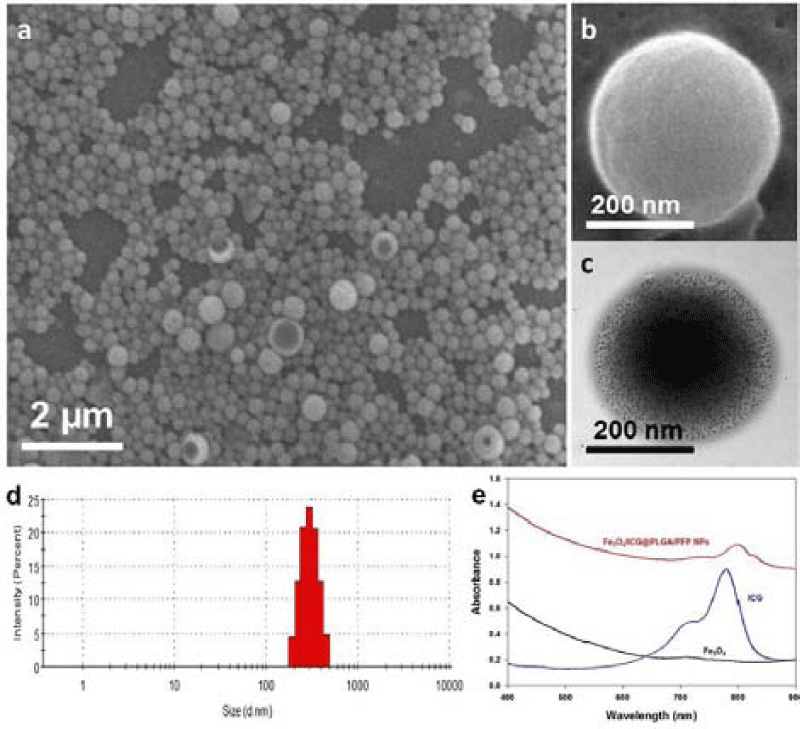 Figure 6: (a,b) SEM images of Fe3O4/ICG@PLGA/PFP NPs (a) 10000×;
scale bar, 2μm; (b) 70000×; scale bar, 200nm); (c) TEM image of
Fe3O4/ICG@PLGA/PFP NPs with a large amount of black Fe3O4 NPs
embedded in the spherical shell (70000×; scale bar, 200 nm); (d) size
distribution of Fe3O4/ICG@PLGA/PFP NPs with the average diameter
of 289.6 ± 67.4 nm; (e) UV-Vis-NIR absorption spectra of Fe3O4/ICG@
PLGA/PFP NPs, free ICG and Fe3O4 NPs. View Figure
Figure 6: (a,b) SEM images of Fe3O4/ICG@PLGA/PFP NPs (a) 10000×;
scale bar, 2μm; (b) 70000×; scale bar, 200nm); (c) TEM image of
Fe3O4/ICG@PLGA/PFP NPs with a large amount of black Fe3O4 NPs
embedded in the spherical shell (70000×; scale bar, 200 nm); (d) size
distribution of Fe3O4/ICG@PLGA/PFP NPs with the average diameter
of 289.6 ± 67.4 nm; (e) UV-Vis-NIR absorption spectra of Fe3O4/ICG@
PLGA/PFP NPs, free ICG and Fe3O4 NPs. View Figure
After in vitro treatment of MCF-7 breast cancer cells with
Fe3O4/ICG@PLGA/PFP NPs and NIR laser irradiation, histology
and electron microscopy confirmed severe damage to the cells
and the formation of many microbubbles with iron particles at the
edge or outside of the microbubbles. In vivo experiments in mice
with MCF-7 tumors demonstrated that Fe3O4/ICG@PLGA/PFP
NPs could achieve tumor ablation upon NIR laser irradiation with
minimal toxicity to non-irradiated tissues. Together, their results
indicate that Fe3O4/ICG@PLGA/PFP NPs can be used as effective
nanotheranostic agents for tumor ablation. Hrebesh M. Subhash,
et al. [36], have been described a functional imaging paradigm
that uses photothermal optical coherence tomography (PT-OCT)
to detect indocyanine green (ICG)-encapsulated biocompatible
poly(lactic-co-glycolic) acid (PLGA) nanoparticles embedded
in highly scattering tissue phantoms with high resolution and
sensitivity. The ICG- loaded PLGA nanoparticles were fabricated
using a modified emulsification solvent diffusion method. With a 20 kHz axial scan rate, PT-OCT based on spectral-domain
interferometric configuration at 1310nm was used to detect
phase changes induced by a 808nm photothermal excitation of
ICG-encapsulated PLGA nanoparticles. An algorithm based on
Fourier transform analysis of differential phase of the spectral
interferogram was developed for detecting the depth resolved
localized photothermal signal. Excellent contrast difference was
observed with PT-OCT between phantoms containing different
concentrations of ICG-encapsulated PLGA nanoparticles. This
technique has the potential to provide simultaneous structural
and molecular-targeted imaging with excellent signal-to-noise
for various clinical applications. Photoacoustic imaging (PAI) is
an emerging biomedical imaging technique that is now coming
to the clinic. It has a penetration depth of a few centimeters
and generates useful endogenous contrast, particularly from
melanin and oxy-/deoxyhemoglobin. Indocyanine green (ICG)
is a Food and Drug Administration-approved contrast agents for
human applications, which can be also used in PAI. It is a small
molecule dye with limited applications due to its fast clearance,
rapid protein binding, and bleaching effect. Edyta Swider et al.
[37], showed that they can label primary human dendritic cells
with the nanoparticles and image them in vitro and in vivo, in
a multimodal manner. This work demonstrates the potential
of combining PAI and 19F MRI for cell imaging and lymph node
detection using nanoparticles that are currently produced at
GMP- grade for clinical use. Photodynamic therapy (PDT) is
emerging as a noninvasive modality for a variety of cancer
treatments [38,39]. In typical PDT, a photosensitizer is activated by external light to produce reactive oxygen species (ROS), which can damage cancer cells [40-42]. For deep-seated tumors, the
efficiency of PDT is decreased due to the limited penetration
depth of external light in biological tissue. This problem has
hindered further development of PDT in widespread clinical
applications. In addition, skin photosensitivity and photothermal
injury are also common concerns from the patients receiving
PDT treatment [43-45]. For the light penetration issue to be
overcome, considerable research has focused on the development
of photosensitizers in the near infrared (NIR) range [46]. Upconversion
nanoparticles combined with photosensitizers have
been explored because they can absorb NIR photons and emit
visible light to activate the photosensitizer for PDT [47,48].
Researchers have explored new approaches, for example, using
Cerenkov radiation to activate a photosensitizer for effective
PDT [49,50]. Alternatively, X-ray is currently widely used in
clinical treatment, and X-ray-activated photodynamic therapy
is another way to overcome the light penetration limitation in
deep-seated tumors [51-53]. The use of an internal light source
is an intriguing solution for the light-penetration problem. For
example, implanting fiber-optic light sources and emitting diodes
could be a viable approach, but it still requires invasive operation
[54-56]. Alternatively, bioluminescence offers an attractive
strategy because of the minimally invasive delivery of enzymes
and substrates. Bioluminescence is a natural phenomenon that
occurs in various organisms, such as marine organisms and some
insects, in which visible light is produced via chemical reactions in
vivo [57,58]. Bioluminescence has been widely used in biological
detection and optical imaging [59-61]. Recently, researchers
have recognized the therapeutic potential of bioluminescence.
Bioluminescence is endogenous fluorescence that can be used
to activate the photosensitizer inside the tumors. This process
is not affected by the light penetration depth of tissue. Wang et
al., reported that luminol has anticancer and antifungal activities
through bioluminescence resonance energy transfer (BRET)
[62]. Moreover, quantum dots have also been combined with
the BRET system for in vivo imaging and PDT in recent reports
[63-65]. Despite these progresses, the BRET-activated ROS
generation and photodynamic effect is largely unexplored. They
show effective photodynamic therapy mediated by the firefly
luciferase bioluminescence system. Biodegradable poly (lacticco-
glycolic acid) (PLGA) nanoparticles were doped with the
photosensitizer Rose Bengal (RB). The PLGA-RB nanoparticles
were then conjugated with luciferase, which produced a
fluorescent signal in the presence of the luciferin substrate.
The endogenous bioluminescence served as a light source to
activate the photosensitizer for ROS generation (Figure 7).
They evaluated cell toxicity, cellular fluorescence imaging, and
therapeutic effect in cellular assays, which indicated that cancer
cells were destroyed effectively. The antitumor effect of BRETPDT
was further investigated in tumor-bearing ICR mice, which
showed that tumor growth was significantly inhibited. This study
provides a promising approach for phototherapy of deep-seated
tumors in the absence of external excitation.
Multifunctional biodegradable and biocompatible poly lacticco-
glycolic acid (PLGA) nanoparticles loaded with indocyanine green (ICG) as an optical-imaging contrast agent for cancer
imaging and as a photothermal therapy agent for cancer treatment.
To improve the efficacy of PTT, poly (lactide co-glycolide) (PLGA)
nanoparticles (NPs) encapsulating superparamagnetic iron
oxide (Fe3O4), indocyanine green (ICG), and perfluoropentane
(PFP) as synergistic agents for NIR laser-induced PTT. ICG after
administered intravenously will be readily bound with blood
proteins and hence leads to only 2 ± 4 min of plasmatic half-life.
Among various pharmaceutical polymers, poly (lactic-co-glycolic
acid) (PLGA) is the copolymer of poly (lactic acid) and poly
(glycolic acid) and is one of the best defined biomaterials with
FDA approval for drug encapsulation due to its biocompatibility,
biodegradability, and controllability for drug release.
 Figure 1: Observation of PGLA microspheres by SEM. View Figure
Figure 1: Observation of PGLA microspheres by SEM. View Figure
 Figure 2: TEM image of presenting of morphology of PIN. View Figure
Figure 2: TEM image of presenting of morphology of PIN. View Figure
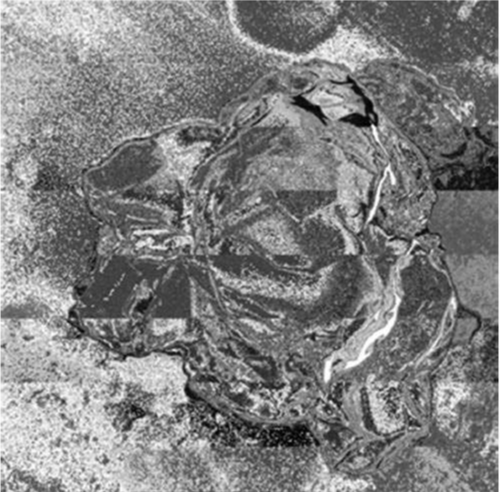 Figure 3: The SEM image of the nGO. View Figure
Figure 3: The SEM image of the nGO. View Figure
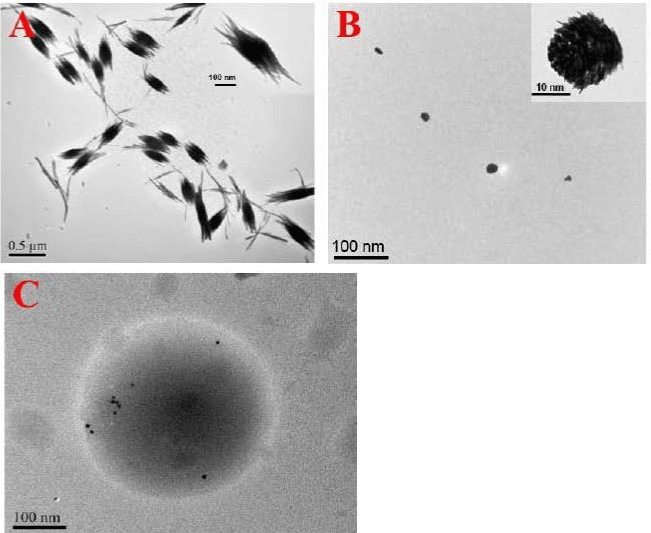 Figure 4: The TEM images of (A) Nanorod-Bundled WO, (B) Granulated. View Figure
Figure 4: The TEM images of (A) Nanorod-Bundled WO, (B) Granulated. View Figure
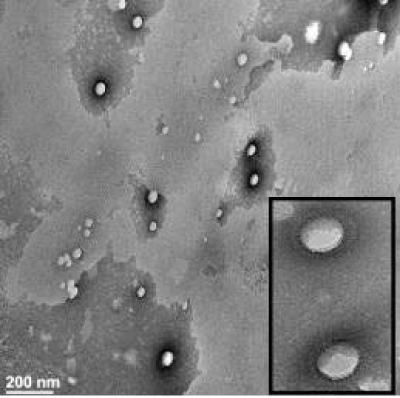 Figure 5: TEM image of Croc/LP-hybrid-NP. View Figure
Figure 5: TEM image of Croc/LP-hybrid-NP. View Figure
 Figure 6: (a,b) SEM images of Fe3O4/ICG@PLGA/PFP NPs (a) 10000×;
scale bar, 2μm; (b) 70000×; scale bar, 200nm); (c) TEM image of
Fe3O4/ICG@PLGA/PFP NPs with a large amount of black Fe3O4 NPs
embedded in the spherical shell (70000×; scale bar, 200 nm); (d) size
distribution of Fe3O4/ICG@PLGA/PFP NPs with the average diameter
of 289.6 ± 67.4 nm; (e) UV-Vis-NIR absorption spectra of Fe3O4/ICG@
PLGA/PFP NPs, free ICG and Fe3O4 NPs. View Figure
Figure 6: (a,b) SEM images of Fe3O4/ICG@PLGA/PFP NPs (a) 10000×;
scale bar, 2μm; (b) 70000×; scale bar, 200nm); (c) TEM image of
Fe3O4/ICG@PLGA/PFP NPs with a large amount of black Fe3O4 NPs
embedded in the spherical shell (70000×; scale bar, 200 nm); (d) size
distribution of Fe3O4/ICG@PLGA/PFP NPs with the average diameter
of 289.6 ± 67.4 nm; (e) UV-Vis-NIR absorption spectra of Fe3O4/ICG@
PLGA/PFP NPs, free ICG and Fe3O4 NPs. View Figure
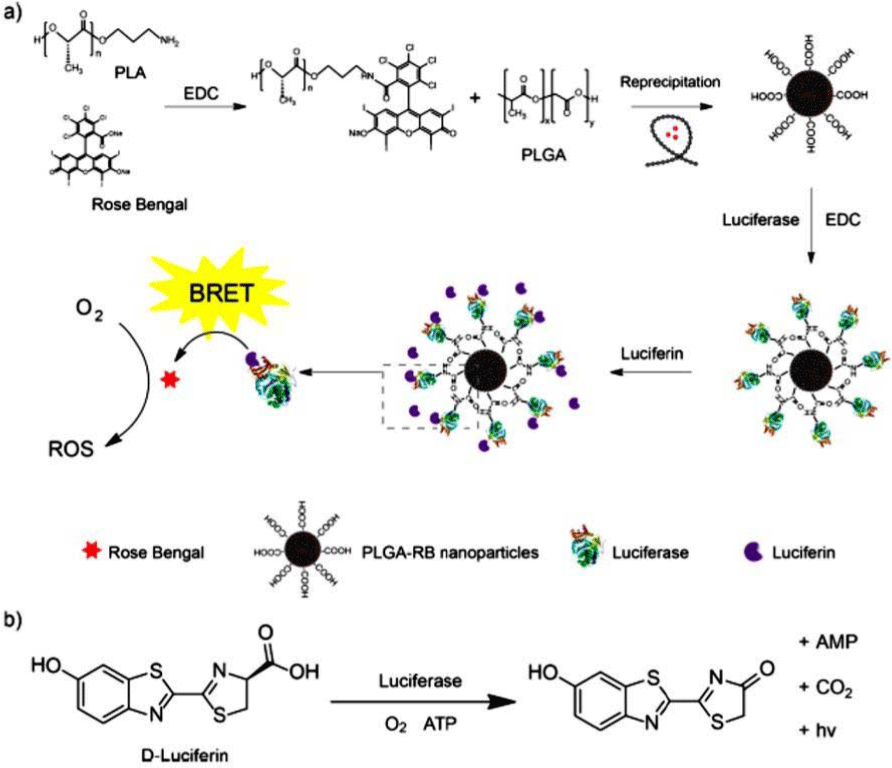 Figure 7: (a) Schematic illustration of the preparation of PLGA-RB
nanoparticles. (b) Firetly luciferase catalyzing the oxidation reaction
of the native substrate D- luciferin for photon emission. View Figure
Figure 7: (a) Schematic illustration of the preparation of PLGA-RB
nanoparticles. (b) Firetly luciferase catalyzing the oxidation reaction
of the native substrate D- luciferin for photon emission. View Figure