Case Report | Volume 2 - Issue 1 | Article DOI : 10.36876/smjnkd.1011
Download PDF
Diana Ramona Dragomir¹ and Dorin Dragoș¹˒²*
¹Nephrology Clinic, University Emergency Hospital Bucharest, Romania
²Department of Medical Semiology, “Carol Davila” University of Medicine and Pharmacy, Bucharest, Romania
Corresponding Author:
Dorin Drago?, Department of Medical Semiology , Carol Davila University of Medicine and Pharmacy, Nephrology Clinic, University Emergency Hospital Bucharest, Romania, Tel: +40 21 318 0719; Email: dordrag@drdorindragos.ro
Keywords
Renal Infarction; Mitral Valve Endocarditis; Aortic Valve Endocarditis; Rheumatic Mitral Valve Disease; Rheumatic Aortic Valve Disease; Splenic Infarction
Abstract
Patients with endocarditis may have various forms of renal impairment; the most commonly encountered being bacterial infection-associated glomerulonephritis and renal infarction by septic emboli.
This is the case of a 68-years-old patient who presented with acute left upper limb ischemia and right lumbar pain. The history revealed acute rheumatic fever in childhood with cardiac involvement (mitral and aortic valve disease, permanent atrial fibrillation), chronic kidney disease and multiple episodes of acute right upper limb ischemia (despite adequate oral anticoagulation) treated conservatively. Thromboendarterectomy was performed in the vascular surgery department. Afterwards the patient was transferred to the nephrology department due to the suspicion of renal infarction. The raising creatinine indicated an acute kidney injury on top of the chronic kidney disease. Transesophageal echocardiography identified images suggestive of vegetation on mitral and aortic valves, spontaneous contrast in the left atrium but without intracavitary thrombosis, fissured atheromatous plaque on the ascending thoracic aorta. As possible causes of renal impairment were considered: renal infarction by thromboemboli originating in the left atrium or by septic emboli from valve vegetation, endocarditis-associated Glomerulonephritis and renal atheroembolism. Renal scintigraphy provided no useful information. Computed tomography without contrast did not reveal areas of renal infarction. The patient refused both Computed tomography angiography (for fear of contrast nephrotoxicity) and valve replacement surgery. Clinical evolution was stationary under antibiotherapy (for endocarditis), high dose statin, besides the previously prescribed treatment with oral anticoagulants, beta blockers, angiotensin converting enzyme inhibitors, dihydropyridine, calcium channel blockers digitalis.
Citation
Dragomir DR and Drago? D. Acute Kidney Injury Associated With Simultaneous Mitral and Aortic Valve Endocarditis, Atrial Fibrillation and Acute Limb Ischemia. J Nephrol Kidney Dis. 2018; 2(1): 1011.
Background
Bacterial endocarditis may have an acute or subacute clinical presentation. The prototypic lesion is the vegetation which is a mass of fibrin, platelets, inflammatory cells, bacterial colonies and it involves the heart valves and also the low pressure side of a ventricular septal defect, mural endocardium damaged by aberrant jets of blood or foreign bodies and intracardiac devices.
The incidence of community-acquired native valve Infective Endocarditis (IE) has remained unchanged in the last three decades. Population studies indicate 3 to 9 cases per 100,000 persons in industrialized countries. The male:female case ratio is more than 2:1. There is a decline in the number of cases associated with rheumatic heart disease (which now accounts for <10% of cases in industrialized countries) and an increase in cases associated with prosthetic valves, intracardiac devices, unrepaired cyanotic congenital heart diseases. Other risk factors include a history of IE, age-related degenerative valvular lesions, hemodialysis and coexisting conditions such as diabetes, human immunodeficiency virus infection and intravenous drug use. The heart lesion found to predispose most frequently to IE is mitral valve prolapse. Staphylococci and streptococci account for 80% of cases of IE, with staphylococci currently the most common pathogens in hospital-acquired IE and streptococci in community acquired and native valve IE [1].
Recent reports have shown that glomerulonephritis is present in 26% of patients with heart valve infection and renal involvement, localized infarcts in 31% of the cases, many of which septic. Interstitial nephritis due to antibiotic therapy and cortical necrosis are the two other factors of renal impairment in this setting.
Case presentation
We describe the case of a 68-years-old male patient who presents with acute left upper limb ischemia and right lumbar pain which appeared 8 hours before the presentation to the emergency room. Lab tests revealed retention of nitrogenous waste products, INR within therapeutic range under chronic oral anticoagulation. Thromboendarterectomy was performed in the vascular surgery clinic. A suspicion of renal infarction was raised; consequently the patient was transferred to our clinic for evaluation.
The history revealed multiple episodes of acute right upper limb ischemia treated conservatively, acute rheumatic fever in childhood with cardiac involvement (mitral and aortic valve disease, permanent atrial fibrillation) and chronic kidney disease (in October 2016 creatinine was 1,9 mg/dL, (eGFR = 35 mL/min/1.73m2 CKD EPI) versus 3,98 mg/dL at the moment of presentation).
On physical examination the temperature was 36.7°C, the oxygen saturation 97% while breathing ambient air, the blood pressure 155/80 mmHg, the pulse rate 68 beats per minute, irregularly irregular cardiac sounds, mitral area systolic murmur (but no opening snap or diastolic murmur), aortic area systolic murmur (but no diastolic murmur), bilateral weak dorsalis pedis pulse, partial edentation, hepatomegaly, diuresis 2000 mL/24 hours, normal urine on macroscopic examination; the physical examination was otherwise normal.
Lab tests revealed
High white blood cells count, neutrophilia, prothrombin-time/ international normalized ratio within therapeutic range under chronic oral anticoagulation, moderately elevated inflammatory markers(fibrinogen 503 mg/dL, Erythrocyte Sedimentation Rate (ESR) 60 mm/h, C-reactive protein 9,69 mg/L), renal insufficiency (creatinine 3,98 mg/dL, urea 123 mg/dL), mild hypocalcemia (7,9 mg/dL), LDH 2 folds the upper limit of the normal range (471 U/L), mild hypertriglyceridemia (172 mg/dL), cholesterol within normal range (197 mg/dL) but high LDL cholesterol (126 mg/dL), qualitatively positive determination for rheumatoid factor; urinalysis: urinary sediment with white cells, red cells, negative urine culture, proteinuria 0.5 g/24 hours; phosphate, plasma proteins and albumin, ALT, AST, C3 fraction of complement, ASO were within the normal range; the patient tested negative for B, C hepatitis.
Electrocardiogram showed atrial fibrillation
Ultrasonography of the abdomen revealed right kidney of normal size but with multiple cysts, the largest with a diameter of about 45 mm, atrophic left kidney, without images suggestive of renal infarction.
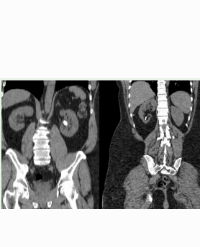
We decided to perform Computed Tomography(CT) scan without intravenous contrast material – it revealed right kidney with irregular contour, intrarenal arterial calcifications, normal cortical index, with several cortical cysts, the largest of 60/45 mm, small left kidney with low cortical index, intrarenal arterial calcifications, fewer cysts, the largest of 16 mm (Figure 1),

Figure 1: CT - kidneys with arterial calcifications. Left-pointing arrow: arterial calcification in the left kidney; right-pointing arrow: arterial calcification in the right kidney. Upward poiniting arrow: cyst in the right kidney.
normal-sized and irregularly contoured spleen with a hypodense area (in the posterior half) of about 30 mm (Figure 2), slightly overall enlarged liver.

Figure 2: CT - splenic infarction. Upward-pointing arrow: splenic infarction; right-pointing arrows: cysts in the right kidney.
We decided to perform renal scintigraphy due to the high suspicion of renal infarction after the CT examination revealed an image suggestive of splenic infarction. The scintigraphy pointed out bilateral normal renal perfusion but with reduced amplitude in the left kidney, functional compensatory right kidney, with participation in global function about 78%, hypofunctional left kidney with participation in global function about 22%.
We also performed transthoracic echocardiography due to the suspicion of intracavitary thrombi in the left atrium- the evaluation revealed rheumatic mitral and aortic valve disease, severe dilation of left atrium without images suggestive of intracavitary thrombi and raised the suspicion of IE of mitral and aortic valves.
Transesophageal echocardiography showed dilated left atrium with intense spontaneous contrast but apparently without thrombosis, dilated left auricle, intense spontaneous contrast (Figures 3 and 5),

Figure 3: TEE - vegetation on atrial side of the posterior mitral valve. Downward-pointing arrow: vegetation; right-pointing arrow: limited opening of the stenotic mitral valve; left-pointing black arrow: dilated left auricle with spontaneous contrast.

Figure 5: TEE -vegetation on the aortic valve. Downward-pointing yellow arrow: vegetation on the aortic valve (little rounded hipoechogenic mass); right-pointing yellow arrow: calcified aortic cusps; left-pointing black arrow: dilated left auricle with spontaneous contrast. LA = Left Atrium.
apparently intact atrial septum, grossly deformed mitral valve, with calcifications on both leaflets but also with images suggestive of vegetation on the anterior surface of posterior leaflet (Figure 3), moderate to severe regurgitation by mixed mechanism (ring dilation and degenerative changes), aortic valve-tricuspid, stenotic (Figure 4),

Figure 4: TEE - turbulent systolic flow through the stenotic aortic valve. Downward-pointing black arrow: turbulent systolic flow through the stenotic aortic valve; right-pointing arrow: turbulent flow in the ascending aorta; left pointing arrow: laminar systolic flow in the left ventricular ejection tract. LV = Left Ventricle, RV = Right Ventricle, LA = Left Atrium, Ao = Aorta.
grossly deformed with large calcifications but also with images suggestive of vegetation on the right coronary leaflet and non-coronary leaflet (Figure 5), secondary regurgitation (Figure 6),

Figure 6: TEE - regurgitant flow from the aortic valve (right-pointing arrow). LV = Left Ventricle, RV = Right Ventricle, LA = Left Atrium, Ao = Aorta.
fibrotic tricuspid valve, with moderate regurgitation, ascending aorta with a calcified atheromatous plaque 38 cm from the origin (Figure 7) and spontaneous contrast in the lumen.

Figure 7: TEE -calcified atheromatous plaque in the ascending aorta (downward-pointing arrow).
At this moment a CT angiography was proposed but the patient declined this investigation because of the risk of additional degradation of renal function induced by the iodine contrast substance.
An eye exam was performed - bilateral stage 2 hypertensive retinopathy and changes suggestive of venous branch thrombosis.
Three blood culture samples yielded negative results.
Clinical evolution was stationary under antibiotherapy (for IE), high dose statin, besides the previously prescribed treatment with oral anticoagulants, beta blockers, angiotensin converting enzyme inhibitors, dihydropyridine calcium channel blockers and digitalis.
Discussion
We describe a case of subacute bacterial endocarditis- according to the Duke criteria: one major clinical criteria (transesophageal echocardiography evaluation) and three minor criteria (predisposing heart disease, embolism by septic emboli in the left thoracic limb and spleen and positive rheumatoid factor). Nonetheless blood cultures were negative. Blood-culture-negative IE raises significant diagnostic and treatment challenges [2].
The patient also had permanent atrial fibrillation with dilated left atrium. Transesophageal echocardiography revealed dilated left atrium with intense spontaneous contrast (despite INR within therapeutic range under chronic oral anticoagulation) and therefore with increased risk of intracavitary thrombus formation constituting a possible thromboembolic source.
Kidney biopsy was contraindicated: absolutely for the right kidney (multiple large cysts) and relatively to absolutely for the left kidney (small size, multiple cysts) [3].
Interesting about this case are the multiple etiological possibilities for the renal damage, each with strong arguments both for and against. Here are the most prominent:
Renal infarction
It occurs in the setting of arterial or venous thrombosis or arterial embolism, heart-related factors (atrial fibrillation, intracavitary thrombus, IE and valvular heart disease) being the main causes [4]. It can involve the entire kidney or small areas of the cortex or medulla. Clinical presentation is characterized by loin, flank or abdominal pain, nausea and/or vomiting, transient or accelerated hypertension and oliguria/anuria (if both kidneys are involved or in the case of a single functioning kidney, but also sometimes in unilateral artery occlusion, supposedly due to contralateral renal artery reflex vasospasm [5]). Lab tests usually reveal signs of tissue damage (increased lactate dehydrogenase and transaminases), renal dysfunction may or may not be present, and urinalysis demonstrates hematuria and proteinuria [6-9]. Interestingly, hematuria is characteristic for and suggestive of renal infarction, but is not mandatory for the diagnosis [10]. In the presence of left-sided IE, renal embolization may represent either a complication or a diagnostic clue [11]. The suspicion of renal infarction should be raised especially when flank pain appears in a patient at risk for thromboembolism. The diagnosis is even more probable it hematuria, leukocytosis and increased LDH are accompanying features [12].
In this case the suspicion of renal infarction by thromboemboli originating in the left atrium or by septic emboli from valve vegetation was supported by right lumbar pain at admission in the hospital, retention of nitrogenous waste products, increased lactate dehydrogenase, microscopic hematuria, subnephrotic range proteinuria, cardiac source, previous thromboembolic events in other territories (upper limb) the existence of a cardiac source (valvular vegetations, spontaneous contrast in the left atrium). However there were no images suggestive of renal infarction on renal scintigraphy, but this was performed three weeks after the flank pain episode. The CT scan was performed without contrast (for fear of aggravating the renal dysfunction), therefore it had little relevance for the diagnosis of renal infarction.
This is the etiology we most favor because it is the only one explaining the patient’s presenting complaints and there was no decisive counter-argument. Unfortunately the definitive imagistic proof of renal infarction was also missing: CT was performed without contrast because of concerns about further compromising kidney function, CT angiography was refused by the patient for the same reason and kidney scintigraphy was performed only after a considerable delay (for logistic reasons).
Endocarditis-associated Glomerulonephritis
The clinical presentation of Endocarditis-Associated Glomerulonephritis (EAGN) is not uniform. Recent data established acute renal failure as by far the most common presentation (~80% of cases) with acute nephritic syndrome, rapidly progressive Glomerulonephritis and nephrotic syndrome in almost equal proportions in the rest of the cases [13]. The extent of glomerular lesions grossly correlates with the severity of clinical findings, focal glomerulonephritis being associated with subnephrotic proteinuria, microscopic hematuria and pyuria (and only rarely with kidney dysfunction), while diffuse glomerulonephritis resulting in micro-or-macroscopic hematuria, nephrotic-range proteinuria and renal insufficiency [14,15]. In some series mesangioproliferative glomerulonephritis was the dominant lesions [16]. In the past crescentic glomerulonephritis was considered an occasional occurrence [15,17]. However, in the latter years necrotizing and crescentic glomerulonephritis seems to be the most common biopsy finding [13]. A classical finding was hypocomplementemia [18,19], but it lacks specificity and seems to be less frequently encountered than previously thought [13]. In the past, complement activation was considered to follow mostly the classical pathway [19,20] and only in some cases (especially associated with staphylococcal infections) alternate pathway [19]. Nowadays, alternate pathway of complement activation appears to be the most prevalent one (low C3 level) probably linked to the higher prevalence of Staphylococcus strains in the etiology of endocarditis-associated glomerulonephritis [13]. The serologic findings are nonspecific, including mixed cryoglobulins and rheumatoid factor [21,18] but also ANCA and ANA [22,13]. The endocarditis induced renal lesion may be quite complex [23], but this is unusual.
Several findings in the present case (inflammatory markers, positive rheumatoid factor and microscopic hematuria) may be the consequence of an EAGN but none of them is specific enough. Even the inflammatory markers and rheumatoid factor are the consequence of endocarditis per se (and not of EAGN), therefore they cannot be used as arguments for an EAGN, nor can be used the normal C3 complement fraction, as complement level may be either normal or decreased in EAGN with roughly the same probability [13]. Moreover EAGN cannot explain flank pain.
Renal Atheroembolism
Aortic atherosclerotic plaques are an important source of emboli, leading to cerebral, extremity or visceral embolization. Embolic events can occur spontaneously or can be induced by interventions including vascular surgery [24]. In our patient the aortic atherosclerotic plaque identified at transesophageal echocardiography may have been a source of spontaneous renal atheroembolism, especially after the procedure performed in the vascular surgery department.
There is no local manifestation characteristic for Renal AtheroEmbolism (RAE). The signs of peripheral ischemia (livedo reticularis, isolated toe cyanosis and even gangrene) are relatively characteristic, although the range of reported incidence in RAE is wide (35-90%) [25,26] Such signs were absent in our patient. Two other characteristic manifestations of RAE, eosinophiluria and eosinophilia [25-28], were also absent in our patient.
Other manifestations of RAE are those of systemic inflammation: fever, myalgias, headaches, and weight loss [25], leukocytosis, elevated inflammatory markers (ESR, C-reactive protein) [27,29,26]. Indeed our patient had leukocytosis and increase inflammatory markers, but they lack specificity. The complement levels may be decreased in RAE; by contrast, they were normal in our patient, but this finding cannot be used either for or against an RAE diagnostic, as it is neither sensitive, nor specific [26,27,29]. In a minority of cases, urinalysis may include granular and hyaline casts (~40%) (Absent in our patient), and less often (<30%) microscopic hematuria and pyuria [25], while more than half of the patients have some degree of proteinuria (present in our patient), sometimes even in the nephrotic range [26,25].
Hence, while renal infarction is the most probable explanation for the patient’s symptoms, RAE is the least probable.
There are several case reports in literature of aortic [30,31] or mitral [32] valve endocarditis associated with renal infarction. However we couldn’t find any with simultaneous aortic and mitral valve endocarditis.
Conclusion
In the compex setting of mitral and/or aortic valve disease, aortic atheromatosis and an endovascular procedure, the most probable cause of an acute kidney injury are: renal infarction due to thromboemboli originating in the left atrium or to septic emboli from valvular vegetations, endocarditis-associated glomerulonephritis, and renal atheroembolism. Embolic events to other territories (such as limbs or spleen) may be additional arguments for a renal infarction.
References
1. Hoen Bruno, Xavier Duval. “Infective Endocarditis.” New England Journal of Medicine. 2013; 368:1425-1433.
2. Hitzeroth J, Beckett N, Ntuli P. “An Approach to a Patient with Infective Endocarditis.” South African medical journal = Suid-Afrikaanse tydskrif vir geneeskunde. 2016; 106: 145-150.
3. Skorecki Karl, Glenn M Chertow, Philip A Marsden, Maarten W Taal, Alan S LYu. Brenner & Rector’s the Kidney. 2016.
4. Huang CC, Chen WL, Chen JH, Wu YL, Shiao CJ. “Clinical Characteristics of Renal Infarction in an Asian Population.” Ann Acad Med Singapore. 2008; 37: 416-420.
5. Levin M, Nakhoul F, Keidar Z, Green J. “Acute Oliguric Renal Failure Associated with Unilateral Renal Embolism: A Successful Treatment with Iloprost.” American journal of nephrology.1998; 18:444-447.
6. Cheng, Kuo-Liang, Shiou-Shan Tseng, Der-Cherng Tarng. “Acute Renal Failure Caused by Unilateral Renal Artery Thromboembolism.” Nephrology Dialysis Transplantation. 2003; 18: 833-835.
7. Huang, Chien-Cheng . “ED Presentations of Acute Renal Infarction.” The American journal of emergency medicine. 2007; 25:164-169.
8. Bourgault Marie, Philippe Grimbert, Catherine Verret, Jacques Pourrat, Michel Herody, Jean Michel Halimi, Alexandre Karras. “Acute Renal Infarction: A Case Series.” Clinical journal of the American Society of Nephrology: CJASN. 2013; 8: 392-398.
9. Oh, Yun Kuy. “Clinical Characteristics and Outcomes of Renal Infarction.” American journal of kidney diseases : the official journal of the National Kidney Foundation. 2016; 67: 243-250.
10. Huang, Chien-Cheng. “Renal Infarction without Hematuria: Two Case Reports.” The Journal of emergency medicine. 2006; 30: 57-61.
11. Menozzi Guido, Valeria Maccabruni, Ermanno Gabbi. “Left Kidney Infarction in a Patient with Native Aortic Valve Infective Endocarditis: Diagnosis with Contrast-Enhanced Ultrasound.” Journal of Ultrasound. 2013; 16:145-146.
12. Korzets Z, Plotkin E, Bernheim J, Zissin R. “The Clinical Spectrum of Acute Renal Infarction.” Isr Med Assoc J. 2002; 4: 781-784.
13. Boils Christie L, Samih H Nasr, Patrick D Walker, William G Couser, Christopher P Larsen. “Update on Endocarditis-Associated Glomerulonephritis.” Kidney international. 2015; 87:1241-1249.
14. Gutman RA, Striker GE, Gilliland BC, Cutler RE. “The Immune Complex Glomerulonephritis of Bacterial Endocarditis.” Medicine. 1972; 51:1-25.
15. Neugarten J, Baldwin DS. “Glomerulonephritis in Bacterial Endocarditis.” The American journal of medicine. 1984. 77: 297-304.
16. Demin AA, Drobysheva VP. “[Kidney involvement in infectious endocarditis (hematuria)].” Terapevticheskii arkhiv. 1991; 63:121-125.
17. Daimon S, Mizuno Y, Fujii S, Mukai K, Hanakawa H, Otsuki N, et al. “Infective Endocarditis-Induced Crescentic Glomerulonephritis Dramatically Improved by Plasmapheresis.” American journal of kidney diseases : the official journal of the National Kidney Foundation. 1998; 32: 309-313.
18. Pelletier L LJr, Petersdorf R G. “Infective Endocarditis: A Review of 125 Cases from the University of Washington Hospitals, 1963-72.” Medicine. 1977; 56: 287-314.
19. O’Connor DT, Weisman MH, Fierer J. “Activation of the Alternate Complement Pathway in Staph. Aureus Infective Endocarditis and Its Relationship to Thrombocytopenia, Coagulation Abnormalities, and Acute Glomerulonephritis.” Clinical and experimental immunology. 1978; 34: 179-187.
20. Kauffmann RH, Thompson J, Valentijn RM, Daha MR, Van Es LA. “The Clinical Implications and the Pathogenetic Significance of Circulating Immune Complexes in Infective Endocarditis.” The American journal of medicine. 1981; 71: 17-25.
21. Hurwitz D, FP Quismorio, GJ Friou. “Cryoglobulinemia in Patients with Infectious Endocarditis.” Clinical & Experimental Immunology. 1975; 19: 131-141.
22. Subra JF, Michelet C, Laporte J, Carrere F, Reboul P, Cartier F, et al. “The Presence of Cytoplasmic Antineutrophil Cytoplasmic Antibodies (C-ANCA) in the Course of Subacute Bacterial Endocarditis with Glomerular Involvement, Coincidence or Association?” Clin Nephrol. 1998; 49: 15-18.
23. Baglin A. “[Prolonged anuria during bacterial endocarditis].” Annales de medecine interne. 1975; 126: 241-250.
24. Scolari Francesco, Pietro Ravani. “Atheroembolic Renal Disease.” Lancet (London, England). 2010; 375: 1650-1660.
25. Fine M J, Kapoor W, Falanga V. “Cholesterol Crystal Embolization: A Review of 221 Cases in the English Literature.” Angiology. 1987; 38: 769-784.
26. Scolari F. “Cholesterol Crystal Embolism: A Recognizable Cause of Renal Disease.” American journal of kidney diseases : the official journal of the National Kidney Foundation. 2000; 36:1089-1109.
27. Lye WC, Cheah JS, Sinniah R. “Renal Cholesterol Embolic Disease. Case Report and Review of the Literature.” American journal of nephrology. 1993; 13: 489-493.
28. Belenfant X, Meyrier A, Jacquot C. “Supportive Treatment Improves Survival in Multivisceral Cholesterol Crystal Embolism.” American journal of kidney diseases : the official journal of the National Kidney Foundation. 1999; 33: 840-850.
29. Thériault J, Agharazzi M, Dumont M, Pichette V, Ouimet D, Leblanc M. “Atheroembolic Renal Failure Requiring Dialysis: Potential for Renal Recovery? A Review of 43 Cases.” Nephron. Clinical practice. 2003; 94: c11-18.
30. Shoji Sunao. “[Renal infarction with renal abscess onset due to infective endocarditis: a casereport].” Hinyokika kiyo. Acta urologica Japonica. 2008; 54: 221-223.
31. Zakaria R, Forsyth V, Rosenbaum T. “A Rare Case of Renal Infarction Caused by Infective Endocarditis.” Nat Rev Urol. 2009; 6: 568-572.
32. Nakayama T, Yokoyama M, Saito K, Takenaka S, Kubo Y, Iimura Y, et al. “[Case of infective endocarditis diagnosed with renal infarction].” Nihon Hinyokika Gakkai zasshi. The japanese journal of urology. 2009; 100: 504-507.