Research Article | Volume 3 - Issue 1 | Article DOI :
Download PDF
Arda Kiani¹, Kimia Taghavi², Maryam Esmaeilzadeh², Adnan Khosravi³, Sharareh Seifi⁴, and Atefeh Abedini²*
¹Tracheal Diseases Research Center, Shahid Beheshti University of Medical Sciences, Iran
²Chronic Respiratory Diseases Research Center, Shahid Beheshti University of Medical Sciences, Iran
³Tobacco Prevention and Control Research Center, Shahid Beheshti University of Medical Sciences, Iran
?Medical Hematology/Oncology, National Institute of Tuberculosis and Lung Disease (NITLD), Iran
Corresponding Author:
Atefeh Abedini, Chronic Respiratory Diseases Research Center, National Research Institute of Tuberculosis and Lung Diseases (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran, Tel: 1956944413; Email: dr.abedini110@sbmu.ac.ir
Keywords
Malignant pleural effusion; Intrapleural chemotherapy; Cisplatin
Abstract
Background: Malignant pleural effusion is a common problem in patients with advanced malignancies and compromised the short-lived survival of these patients. These effusions can be resistant to the treatment such as systemic chemotherapy, pleurodesis with sclerosantagents and recurrent drainage. Therefore, new therapeutic options are needed. The purpose of this study was to evaluate the effectiveness of intrapleural chemotherapy with Cisplatin for the management of MPE in lung, breast and mesothelioma cancers.
Materials and methods: Twenty-one cancer patients with MPE were enrolled in this study. Cisplatin was injected through a catheter at a dose of 30mg /m2, and this procedure was performed 3 times at intervals of two weeks. Patients were evaluated for side effects and responses to the treatment every two weeks and one month after the last treatment.
Results: Among the assessable 18 patients, complete response and partial response were 9 (50%) and 4 (22.2%) patients, respectively (overall response rate 72.2%). Dyspnea was improved in 13 (72.2%) patients and had no change in 5 (27.8%) patients. One patient did not refer after the first intrapleural injection. Also two patients died during the study. None of the patients had side effects of grade 3 and 4.
Conclusion: The results of this trial study showed that using of Intrapleural Chemotherapy with cisplatin in the management of patients with MPE in lung, breast and mesothelioma cancers is effective and safe.
Introduction
Malignant Pleural Effusion (MPE) occurs in most patients with advanced cancer [1] and accounts for 15 to 35 percent of all pleural effusions [2]. Most MPEs are caused by metastases, especially in lung cancer, in more than one-third of cases and in breast cancer in the latter stages [3]. Variety of symptoms such as progressive dyspnea, cough and chest pain reduces the quality of the short-lived survival of these patients [4] and in some cases can be fatal and therefore, it is associated with poor prognosis and significant mortality and morbidity [1]. Despite all treatments such as surgery, chemotherapy and radiotherapy, survival rates in primary pulmonary cancer patient with MPE are low [5]. This effusion usually does not respond to systemic chemotherapy and treatment is mostly symptomatic [6]. In many patients, tubal drainage and pleurodesis are used to control plural effusion by injecting sclerosing agents, but the failure to respond to pleurodesis require frequent necessity for drainage through a chest tube, leading to multiple visits to medical centers and extended hospitalization and greater risk of infection [4-7], thus new approaches in therapy are essential. One of these treatments is Intra Pleural Chemotherapy (IPC) with anticancer drugs that have been studied in interventional therapy. Mentioned treatment is performed locally, so systemic side effects are minimized and the procedure is quite effective [8]. In patients who did not respond to Systemic Chemotherapy or for any reason were not candidates for Chemotherapy, IPC can reduce the accumulation of pleural fluid and relieve the patient’s symptoms and improve their quality of life [9]. One of the new therapeutic interventions of IPC is anticancer drugs, which has been studied in interventional procedures. A study in South Korea showed that IPC with Cisplatin and Cytarabine was effective in the treatment of MPE in Cervical Carcinoma with multiple pulmonary and cerebral metastases [1]. Also, other studies in Japan and China have proven the efficacy of IPC in the treatment of MPE [10]. Accordingly, the present study was designed to evaluate the effect of IPC with cisplatin in the treatment of MPE in an Iranian population.
Methods
Patients selection
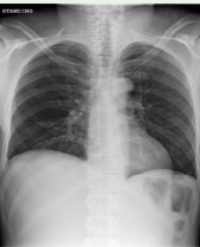
This study was approved by Iranian registry clinical trials (IRCT2017053134256N1). Cancer patients with pleural effusion who referred to Massih Daneshvari Hospital from March 2016 to February 2017 were enrolled in this study. Among them, twenty-one cancer patients with pleural effusion in lung, breast and mesothelium who have positive cytology for malignancy, and have not responded to systemic chemotherapy or have not responded to standard systemic chemotherapy for any reason, were selected. Of all patients, the chest X-ray was performed in order to estimate the initial degree of pleural effusion. All patients gave written informed consent.
Treatment protocol
After insertion of the pleural catheter and dilution of the pleural fluid, cisplatin was injected through a catheter at a dose of 30 mg/m2 , and this procedure was performed 3 times at intervals of two weeks. If pleural effusion was completely resolved after the first or second injection, the injection was not repeated longer and only follow-up was considered. Before each injection, Complete Blood Count (CBC), kidney function test and electrolytes were examined for each patient in order to investigate the side effects. The estimation of the amount of pleural effusion in the chest X-ray and the comparison of each step was made by the radiologist by naked eye. The complete response to treatment was considered when pleural effusion completely disappeared and did not recur after four weeks. The relative response was defined as a decrease in fluid but with no complete disappearance and improvement in the patient’s dyspnea and no accumulation of the fluid within four weeks after the end of the treatment. No response was defined as when the fluid level did not change, or increased within four weeks after the end of the treatment. Out of the 21 patients enrolled in the study, one patient did not refer after the first intrapleural injection, due to dyspnea’s dislocation, and none of analyzes was performed. Also two patients died during the study.
Statistical analysis
The data were collected and categorized into SPSS version 22. Descriptive data are expressed as the mean ± standard deviations. Nonparametric Wilcoxon and Kruskal-Wallis statistical tests were used to determine the effect of IPC on the amount of pleural effusion and dyspnea. P-value <0.05 was considered as significant in all groups.
Result
Patients characteristics
In the period of March 2016 to February 2017, twenty one patients from Massih Daneshvari Hospital were enrolled in this study as shown in (Table 1). Out of 21 patients, 61.9% were men and 38.1% women with mean age of 60.95 years and age range of 47 to 80 years. Ten patients had MPEs as a consequence of primary metastatic lung cancer (stage IV), 6 patients had MPEs due to breast cancer and 2 patients had MPEs owing to mesothelioma. Nineteen patients had a history of systemic chemotherapy but 2 patients did not receive any systemic chemo drug.
Table 1: Patient characteristics.

Drug administration
Out of 18 patients who completed the study, 50% (9 patients) had a complete response to Cisplatin intra-pleural, 22.2% (4 patients) had partial response and 50% had no response (Figure 1).

Figure 1: Frequency of distribution of patients based on response to Intrapleural Cisplatin.
Kruskal Wallis test showed no significant difference in the amount of pleural effusion, before the first injection and in three periods of completed response, partial response, and lack of response (P> 0.05). The comparison between the two groups of pre-intervention and post-intervention is presented in (Table 2) , during three different period and based on the percentage of pleural effusion in chest X-ray, using nonparametric Wilcoxon test statistics .The results of the test showed a significant difference between all the groups (P=0.002).
Table 2: The comparison of the two groups of pre-intervention and postintervention at three different times.

Out of ten male patients who completed the study, 30% (3 patients) had completed response to intra-pleural cisplatin, 20% (2 patients) had partial response and 50% (5 patients) had no response. Out of eight female patients who completed the study, 75% (6 patients) had completed response to intra-pleural cisplatin and 25% (2 patients) had no response. At the end of study, in response to intra-pleural cisplatin, out of 10 patients with lung cancer 4 cases had complete response, 2 cases had a partial response and 4 cases had lack of response. Out of 6 cases of breast cancer, 4 patients had complete response and 2 patients had partial response. From 2 cases of mesothelioma, 1 case had complete response and 1 case had no response (Figure 2).

Figure 2: Frequency of distribution of response to Intra-pleural Cisplatin based on the type of primary cancer.
injection, in 4 cases in the second follow-up followed by the second injection and in 3 cases in the third follow-up followed by the third injection. Table 3 shows the comparison of the two groups of preintervention and post-intervention at three different times, based on Borg dyspnea Scale, using the nonparametric Wilcoxon test. The results of the test showed a significant difference in all groups (P< 0.05)
Table 3: The comparison of the two groups (before and after the intervention at three different times) in terms of Dyspnea according to the Borg Scale questionnaire.

Side effects created during treatment
Following treatment anemia has been observed in 2 patients, Thrombocytopenia grade II in 2 patients, Hypomagnesaemia grade I in 4 patients and Hypokalemia grade I in one patient. Hypocalcemia grade I and II was observed in three and one patient, respectively.4.
Discussion
The purpose of this study was to determine the effect of intrapleural cisplatin chemotherapy on the treatment of MPE. MPE is often the result of impaired pleural fluid reabsorption due to mediastinal lymph node obstruction that is responsible for drainage of pleural fluid [11]. Tumors that metastasize to these lymph nodes such as lung, breast and lymphoma cancers are the most common causes of MPE [12]. One of the main therapeutic priorities is the active control of pleural effusion in order to improve the patient’s quality of life [10]. One of these treatments is IPC with anticancer drugs. Cisplatin is a known chemotherapeutic drug with anti-tumor activity [7] that interacts with DNA and cause apoptosis and inhibition of cell growth [13].
In the present study, twenty one cancer patients enrolled for Cisplatin IPC. Out of 18 patients persisting by the end of the study, 9 patients (50%) had completed response, 4 (22.2%) had a partial response and 5 patients (27.8%) had no response to treatment. Based on these results, the total overall response rate of complete response and partial response were 13 cases (72.2 %). Due to cisplatin, none of the patients had hematologic and non-hematologic complications. Nan Du et al. studied the effect of intra-pleural treatment in combination with bevacizumab and cisplatin compared with individual treatment only by cisplatin, in 72 patients with NSCLC. At the end of the study, the overall response rate in the group just receiving cisplatin (control group) was 50 percent.
The response to the treatment was evaluated weekly and through Pleural Sonography (10), while in the present study, the response to treatment was evaluated with chest X-ray. In a study by Figlin R et al. , performed on 46 patients with MPE with cytological confirmation in a variety of solid tumors, intra-pleural cisplatin injection was performed as a single dose of 100 mg/m2 with 1200 mg of cytarabine via chest tube and overall response rate (complete and partial) after three weeks was 49 percent [13].
This is due to the injection that was performed only in one cycle and the overall response rate was lower than the present study, the more general response in this study can be attributed to more frequent intra-pleural injection. In another study by Tetsuro Baba and colleagues, in 8 out of 17 patients with NSCLC, IPC was performed with cisplatin or Adriamycin but not with other IPC. In the group under IPC, the survival rate was 88% but in non-IPC group it was 44% (P = 0.04) and they concluded that IPC is effective in these patients and improves postoperative survival [14]. In terms of improved dyspnea, out of 18 patients examined, 13 cases (72.2%) had improvement in dyspnea and in 5 cases (27.8%) no changes has been observed. In the present study, twenty patients were examined for the presence of any side effects after treatment, no one shows sign of leucopenia or neutropenia, renal dysfunction and hearing impairment but only 2 patients show sign of anemia 2 patients had thrombocytopenia, 1 patient had Hypokalemia, 4 patients had Hypocalcemia, and 4 patients had Hypomagnesaemia. In terms of side effects, in a study by Figlin R et al., a patient was diagnosed with grade III nephrotoxicity, 4 patients had grade III hematologic complications and 5 patients had grade III cardiovascular complications [13].
In a study by Kee Won Kim et al., in 40 patients with NSCLC one cycle of IPC with cisplatin at a dose of 100 mg/m2 and cytarabine at a dose of 1200 mg/m2 was performed via chest tube, and one case of death due to drug toxicity and one case of grade IV hematologic complications have been observed [15]. Two patients had Empyema and wound infections, but no significant renal or hepatic toxicity was observed in any of these patients [15]. The more severe side effects in these two studies can be attributed to the simultaneous combination of cisplatin and cytarabine, and also to the use of a higher dose of cisplatin (100 mg/m2 ). In previous studies consistent with our study, no severe therapeutic side effects such as grade III or IV has been observed. We speculate that since the injection of cisplatin was performed locally and within the pleural, no serious systemic side effects would be expected.
Although due to the low sample size and the lack of uniformity of the patients, it is not possible to compare and accurately evaluate the types of cancers for the response to cisplatin in the intrapleural injection. According to the findings of this study, it is suggested that breast cancer has a better response to therapy than lung and mesothelioma cancer. In conclusion, a larger sample size and matched patients are required in order to compare the response of intrapleural cisplatin injection in patients with lung cancer and breast cancer and mesothelioma cancer.
References
1. Monfared ZE, Khosravi A, Naini AS, Radmand G, Khodadad K. Analysis of Cisplatin-Induced Ototoxicity Risk Factors in Iranian Patients with Solid Tumors: A Cohort, Prospective and Single Institute Study. Asian Pacific Journal of Cancer Prevention. 2017; 18: 753-758.
2. Mirhosseini SM, Fakhri M, Mozaffary A, Lotfaliany M, Behzadnia N, Ansari Aval Z, et al. Risk factors affecting the survival rate in patients with Symptomatic Pericardial Effusion undergoing surgical intervention. Interact Cardiovasc Thorac Surg. 2012; 16: 495-500.
3. Fahimi F, Khodadad K, Amini S, Naghibi F, Salamzadeh J, Baniasadi S. Evaluating the effect of Zingiber Officinalis on Nausea and Vomiting in patients receiving Cisplatin based regimens. Iranian journal of Pharmaceutical Research: IJPR. 2011; 10: 379.
4. Khosravi A, Esfahani-Monfared Z, Karimi SH, Emami H, Khodadad K. Comparing Docetaxel with Gemcitabine as second-line chemotherapy in patients with advanced non-small cell lung cancer: A single institute randomized phase II study. J Cancer Res Ther. 2015; 3: 1-7.
5. Karimi S, Yousefi F, Seifi S, Khosravi A, Nadji SA. No evidence for a role of Merkel cell Polyomavirus in small cell lung cancer among Iranian subjects. Pathology-Research and Practice. 2014; 210: 836-839.
6. Tafsiri E, Darbouy M, Shadmehr MB, Zagryazhskaya A, Alizadeh J, Karimipoor M. Expression of miRNAs in non-small-cell lung carcinomas and their association with clinicopathological features. Tumor Biology. 2015; 36: 1603-12.
7. Jacobs RE, Gu P, Chachoua A. Reactivation of Pulmonary Tuberculosis during Cancer Treatment. International Journal of Mycobacteriology. 2015; 4: 337-340.
8. ShaloamDasari, Paul Bernard Tchounwou. Cisplatin in cancer therapy: Molecular mechanisms of action. Eur J Pharmacol. 2014; 740: 364-378.
9. Miller RP1, Tadagavadi RK, Ramesh G, Reeves WB. Mechanisms of Cisplatin nephrotoxicity. Toxins. 2010; 2: 2490-2518.
10. Nan DU, Xiaosong Li, Fang Li, Hui Zhao, Zhongyi Fan, Junxun Ma, et al. Intrapleural combination therapy with bevacizumab and cisplatin for nonsmall cell lung cancer mediated malignant pleural effusion. Oncology Reports. 2013; 29: 2332-2340.
11. Light RW, Amber D. Malignant Pleural Effusions. In: Pleural Diseases. Williams and Wilkins. 1995; 94: 116.
12. Fredric W, Grannis J, JY Kim, Lily Lai. Fluid complications: Malignant pleural effusion. Oncology Journal. 2015; 73: 213-216.
13. Figlin R, Mendoza E, Piantadosi S, Rusch V. Intrapleural Chemotherapy without Pleurodesis for Malignant Pleural Effusions: LCSG Trial 861. Chest. 1994; 106: 363S-366S.
14. Baba T, Uramoto H, Kuwata T, Takenaka M, Chikaishi Y, Oka S, et al. Intrapleural chemotherapy improves the survival of non-small cell lung cancer patients with positive pleural lavage cytology. Surg Today. 2013; 43: 648-653.
15. Kim KW, Park SY, Kim MS, Kim SC, Lee EH, Shin SY, et al. Intrapleural chemotherapy with cisplatin and cytarabine in the management of malignant pleural effusion. Canserres treat,2004;36:68-71.
Citation
Kiani A, Taghavi K, Esmaeilzadeh M, Khosravi A, Seifi S and Abedini A. Evaluation of Intrapleural Chemotherapy with Cisplatin in Iranian Cancer Patients with Malignant Pleural Effusion. SM J Pulm Med. 2017; 3(1): 1027s1.