Alejandra M. Mena-Gutierrez1, Ryan C. Maves2, Jonathan E. Kelly3, Scott A. Kaczmorski3, William Doares3, Christopher J. Webb4 , Alan C. Farney4, and Robert J. Stratta4*
1Department of Internal Medicine, Section of Nephrology, Wake Forest University School of Medicine, USA
2Department of Internal Medicine, Section of Infectious Disease, Wake Forest University School of Medicine, USA
3Department of Pharmacy, Atrium Health Wake Forest Baptist, USA
4Department of Surgery, Section of Transplantation, Wake Forest University School of Medicine, USA
Corresponding Author:
Robert J. Stratta, Department of Surgery, Section of Transplantation, Wake Forest University School of Medicine, Winston Salem, NC, USA, Tel: (001) 336-716-054
Abstract
The limited supply of available donor kidneys has led to a reexamination of donor selection criteria to meet demand. This reevaluation is also influenced by medical advancements that have allowed for the prompt identification and safe management of donor-transmitted infections without compromising outcomes. Historically, the presence of aspergillosis in a deceased donor has been a contraindication to kidney use in transplantation. However, isolated favorable reports, coupled with successful management of de novo aspergillosis following kidney transplantation, have provided insights regarding the benefits and risks of this strategy. Herein, we report the successful transplantation of a kidney donated after circulatory death from a donor with chronic cavitary pulmonary aspergillosis. Based on literature review and given the benefits of kidney transplantation versus dialysis in the setting of prolonged waiting times, expansion of the donor pool to include kidneys from donors being actively treated for opportunistic infections may be an alternative option to increase utilization and access.
Abstract
The limited supply of available donor kidneys has led to a reexamination of donor selection criteria to meet demand. This reevaluation is also influenced by medical advancements that have allowed for the prompt identification and safe management of donor-transmitted infections without compromising outcomes. Historically, the presence of aspergillosis in a deceased donor has been a contraindication to kidney use in transplantation. However, isolated favorable reports, coupled with successful management of de novo aspergillosis following kidney transplantation, have provided insights regarding the benefits and risks of this strategy. Herein, we report the successful transplantation of a kidney donated after circulatory death from a donor with chronic cavitary pulmonary aspergillosis. Based on literature review and given the benefits of kidney transplantation versus dialysis in the setting of prolonged waiting times, expansion of the donor pool to include kidneys from donors being actively treated for opportunistic infections may be an alternative option to increase utilization and access.
Citation
Stratta RJ, Webb CJ, Jay CL, McCracken E, Garner M et al, (2025) Improved Outcomes Following Simultaneous Pancreas-Kidney Transplantation in the Second Decade of the New Millennium. J Nephrol Kidney Dis 6(3): 5.
INTRODUCTION
Addressing the ongoing shortage of donor organs is one of the most important and difficult problems facing kidney transplantation today. The burden of end stage kidney disease continues to exceed the availability of donor organs in the quest to offer kidney transplantation as the preferred method of renal replacement therapy. The aging of both donor and recipient populations, with increases in associated co-morbidities, has dramatically increased the complexity of decision-making with respect to finding ‘the right kidney for the right recipient.’ The organ procurement community has responded by aggressively pursuing all eligible donors, including donation after circulatory death (DCD) donors as well as those with chronic infections that previously were contraindications to organ donation. In the past decade, the number of DCD donors in the US has increased 5.5-fold (from 1,277 in 2014 to 6,977 in 2024) [1,2]. In 2024, DCD donors accounted for nearly 44% of deceased donors in the US, representing a doubling in the number of DCD kidney donors since 2020. However, more than 5000 kidneys recovered from DCD donors were not transplanted in 2024, taxing resources and potentially missing opportunities for gained quality life-years in appropriately selected recipients [1]. In view of this, we report the successful transplant of a kidney from a DCD donor with chronic cavitary pulmonary aspergillosis.
DONOR HISTORY
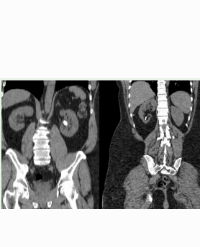
The donor was a Black man in his early forties, height 65 inches (165 cm), weight 179 lbs. (81.4 kg), body mass index 30.2 kg/m2, blood type O, Kidney Donor Profile Index (KDPI) score 51%, with three tattoos. His past medical history was notable for pulmonary sarcoidosis complicated by traction bronchiectasis and a 6-month history of invasive Aspergillus with fungus balls/cavitary lesions present for which he had been treated with oral voriconazole. He additionally had coronary artery disease with prior myocardial infarction and ischemic cardiomyopathy, previous tobacco use, and heavy alcohol use. The donor also had a history of bacterial pneumonia superimposed on aspergillosis, treated with intravenous piperacillin/tazobactam and oral amoxicillin/clavulanic acid in the recent past. He suffered an out-of-hospital hypoxic arrest secondary to massive hemoptysis that was initially managed with cardiopulmonary resuscitation and two rounds of epinephrine with return of spontaneous circulation while being transported to an outlying Emergency Department. An additional cardiac arrest with pulseless electrical activity occurred that responded to one dose of epinephrine. The estimated total “down time” was at least 14 minutes. Following endotracheal intubation and stabilization, he was transferred to the eventual donor hospital, where he was noted to have severe neurological deficits secondary to anoxic ischemic encephalopathy. A transthoracic echocardiogram demonstrated a left ventricular ejection fraction <20%. He was initially managed with vasopressors and inotropic support and was successfully weaned off after 48 hours. He did not require any blood products. He received intravenous voriconazole in addition to empiric broad-spectrum antibacterial therapy. Initial leukocyte count was 22,500/mm3 and terminal leukocyte count was 30,300/mm3. The patient was hospitalized for 9 days and had no overt evidence for disseminated aspergillosis. Multiple imaging studies with contrast (computed tomography [CT] scans of head, chest, abdomen, and pelvis; magnetic resonance imaging of the brain) revealed diffuse ischemic encephalopathy with no masses, multifocal areas of bilateral cavitary pneumonia (Figure 1a, axial view; Figure 1b, coronal view), bullous changes, dense atelectasis, pleural effusions, and ileus. Multiple blood and urine cultures were negative. Bronchoalveolar lavage specimens were significant only for a scant growth of Haemophilus influenzae. Urine output remained above 100 ml/hour. Admission serum creatinine level was 0.77 mg/dl and terminal creatinine level was 0.60 mg/dl. Liver enzymes and coagulation parameters remained normal. Following initial resuscitation and stabilization, there were no further problems with oxygenation, ventilation, or acid-base disturbances. The patient had no signs of neurological recovery, and the family provided consent for withdrawal of life support and subsequently for organ donation. Total warm ischemia time was 23 minutes and functional warm ischemia (from systolic blood pressure and oxygen saturation both <80 to initiation of in situ flush) was 16 minutes. Only the kidneys were recovered, and both kidneys flushed well and appeared normal with no visible parenchymal or vascular abnormalities (no aortic and arterial plaque). Both kidneys were 11.5 x 7-8 x 3 cm in size and had normal anatomy (single arteries, veins, and ureters). Both kidney biopsies exhibited 2% glomerulosclerosis (1/56 and 1/66 glomeruli, respectively) with focal tubular injury, focal inflammation, and no vascular changes. Both kidneys were placed separately on Organ Recovery Systems LifePort® pumps with excellent pump parameters; 4-hour pump numbers for the left kidney were a flow of 152 ml/min with a resistance value of 0.12 mm Hg/ml/min; corresponding pump numbers for the right kidney were a flow of 144 ml/min with a resistance of 0.18 mm Hg/ml/min.

Figure 1a: Chest CT scan of donor, axial view, shows large cavitary lesion in the left upper lobe containing ovoid soft tissue mass surrounded by gas in the cavity. There are also smaller cavitary lesions, extensive consolidative opacities with evidence for traction bronchiectasis, bilateral apical cystic changes, and a small right pleural effusion.
RECIPIENT COURSE
Upon receiving the initial kidney offer from a non-local organ procurement organization, consultation was obtained by the transplant surgeon on-call from our local transplant infectious disease specialist. A decision was made to accept only the left kidney as an open or aggressive offer. We were granted waivers prior to importing the kidney and chose a recipient who was in the top fifty on the Match Run list but not at the top of our list. The recipient is a white man in his early sixties, height 68 inches (172.7 cm), weight 214 lbs. (97.1 kg), body mass index 32.5 kg/m2, blood type O, Estimated Post-Transplant Survival score 86%. End stage kidney disease was secondary to biopsy-proven focal segmental glomerulosclerosis (FSGS). The recipient had been placed on our waiting list 5 months previously and had been receiving hemodialysis for 48 months. He was given general information about the donor including the need to receive anti-microbial treatment post-transplant because of a history of pneumonia in the donor. The recipient had a calculated panel reactive antibody level of 0% and had a human leukocyte antigen mismatch of 2-1-1 with the donor. The virtual crossmatch was negative, and the transplant was initiated upon arrival of the organ to minimize cold ischemia time, which is our standard of practice when the recipient is not sensitized. The subsequent physical crossmatch (by T and B lymphocyte flow cytometry) was negative. Total pump time was 18 hours with a terminal low of 118 ml/min and a terminal resistance value of 0.21 mm Hg/ml/ min. The recipient was in the high-risk category for cytomegalovirus (CMV) transmission (donor seropositive and recipient seronegative). In addition to FSGS, the recipient had a past medical history of type 2 diabetes mellitus, hypertension, asthma, gout, obstructive sleep apnea, dyslipidemia, former tobacco use, multi-vessel coronary artery bypass grafting, peripheral vascular disease, mild chronic leukocytosis (leukocyte count ranging from 11,000 to 12,600 /mm3 with normal differential count), and had undergone a previous robotic prostatectomy for prostate cancer. He had been removed from the transplant waiting list at another center because of co-morbidities and lack of vascular targets for kidney implantation. Prior non-contrast abdominal and pelvic CT scan had revealed severe concentric calcific atherosclerosis of both iliac arterial systems extending from the aortic bifurcation to the femoral vessels. However, imaging had revealed a target relatively free of calcifications in the proximal left common iliac artery.

Figure 1b: Chest CT scan of donor, coronal view, demonstrating bilateral aspergillomas in a background of interstitial lung disease with superimposed bronchopneumoni.
diabetes mellitus, hypertension, asthma, gout, obstructive sleep apnea, dyslipidemia, former tobacco use, multi-vessel coronary artery bypass grafting, peripheral vascular disease, mild chronic leukocytosis (leukocyte count ranging from 11,000 to 12,600 /mm3 with normal differential count), and had undergone a previous robotic prostatectomy for prostate cancer. He had been removed from the transplant waiting list at another center because of co-morbidities and lack of vascular targets for kidney implantation. Prior non-contrast abdominal and pelvic CT scan had revealed severe concentric calcific atherosclerosis of both iliac arterial systems extending from the aortic bifurcation to the femoral vessels. However, imaging had revealed a target relatively free of calcifications in the proximal left common iliac artery. Consequently, a decision was made to place the kidney in the left iliac fossa through a standard retroperitoneal approach. The patient received basiliximab induction therapy (rather than alemtuzumab, which is our standard practice) because of the history of aspergillosis in the donor and prostate cancer in the recipient. The dissection was technically difficult, and assistance from Vascular Surgery was obtained due to calcific atherosclerosis. The total cold ischemia time was 25 hours, operating time was 5.5 hours, and anastomosis time was 37 minutes. A ureteral stent and perinephric drain were placed, and estimated blood loss was 200 ml. The recipient remained hemodynamically stable throughout the procedure, the kidney reperfused well, and a mild diuresis was noted at the end of the procedure. A reperfusion needle biopsy for permanent section was performed, which subsequently showed 0% glomerulosclerosis, minimal interstitial fibrosis and tubular atrophy, and mild arteriosclerosis. The patient experienced delayed graft function and received three hemodialysis treatments in the first 8 days post-transplant; length of initial hospital stay was 4 days. A second dose of basiliximab was given just prior to hospital discharge. Maintenance immunosuppression consisted of tacrolimus (target 12-hour trough levels of 6-9 ng/ml initially), mycophenolic acid 360 mg twice daily, and a prednisone taper from 20 mg/d to 5 mg/d within the first month post-transplant. In addition to peri-operative surgical site prophylaxis, at the time of discharge the recipient was placed on our standard regimen of valganciclovir for 6 months (because of primary CMV exposure) and sulfamethoxazole trimethoprim for at least one year. In place of fluconazole, following consultation with the Infectious Disease team, the recipient was started on oral posaconazole 300 mg daily for 3 months, tacrolimus dosing was adjusted accordingly because of the potential for a drug interaction, and levels of both agents were monitored closely. The maintenance oral tacrolimus dose was reduced to 0.5 mg every other day to achieve trough levels of 6-9 ng/ml for the first 3 months post-transplant. The recipient was followed closely in the Transplant and Infectious Disease clinics. Serial blood and urine cultures, serum1,3-beta-D glucan levels, and Aspergillus galactomannan antigen tests remained negative. Renal function steadily improved and the serum creatinine level stabilized in the 1.1-1.3 mg/dl range by 6 weeks post-transplant. Urine protein/creatinine ratios remained 1000 ng/ml, normal 24-330 ng/ml). The patient was re-admitted twice for evaluation of leukocytosis and Hematology and Infectious Disease consultations were obtained. Multiple cultures and imaging studies (including positron emission tomography and tagged leukocyte scan) failed to reveal any occult evidence for infection other than stranding along the transplant ureter with no evidence of urinary tract infection or retained stent. Flow cytometric analysis of peripheral blood did not identify any aberrant B-cell, T-cell, or blast populations and a next-generation sequencing myeloid neoplasm panel did not detect any clinically significant DNA variants or gene fusions. BK, CMV, JC, and Epstein-Barr viral polymerase chain reaction levels remained undetectable. Interferon-gamma release assay testing was negative. At 6 months post-transplant, both valganciclovir and posaconazole prophylaxis were stopped and tacrolimus dosing was increased to 1.0 mg twice daily. The recipient subsequently underwent a dental extraction for an infected tooth, after which time the total leukocyte count stabilized in the 10,000 to 12,000/mm3 range. He continues to do well at 12 months post-transplant with no localizing or constitutional signs or symptoms. Renal function remains stable (serum creatinine levels 1.2-1.3 mg/dl, estimated glomerular filtration rate 65 ml/min/1.73m2) with tacrolimus trough levels in the 4-6 ng/ml range. Surveillance cultures, viral, and fungal assays remain negative.
Table 1: Literature review of donor-derived aspergillosis following kidney transplantation
|
Authors, year, reference
number, center,
|
Study design, number of cases, findings
|
Outcomes
|
|
Keating, et al., 19969, Hennepin County and the University of Minnesota
|
Case report – 2 kidneys and 1 heart from same donor (who was a liver transplant recipient) developed invasive aspergillosis
|
Both kidney patients survived following
nephrectomy; heart recipient survived following aortic valve replacement and prolonged amphotericin B therapy
|
|
Garrido, et al., 200310, University Hospital, Salamanca, Spain
|
Case report – both kidney recipients from same donor developed iliac artery pseudoaneurysms from Aspergillus species and underwent nephrectomies
|
One patient reconstructed with graft and
subsequent died; other patient survived following iliac artery ligation and prolonged liposomal amphotericin therapy
|
|
Mueller, et al., 20107, University Hospital Zurich, Switzerland
|
Case report – both kidney recipients from same donor
(who was a heart transplant recipient) developed Aspergillus infections; liver recipient treated preemptively with voriconazole with no evidence for subsequent Aspergillus infection
|
One patient underwent nephrectomy and
survived; other patient diagnosed and treated for Aspergillus urinary tract infection with voriconazole for 10 months and maintained graft function
|
|
Shoham, et al., 20108, Washington Hospital Center, Washington, DC
|
Review of 19 cases of invasive fungal infections in 17 patients post commercial (living unrelated donor) kidney transplant including 12 with Aspergillus species
|
Invasive mold infection present in allograft in
35% of cases; 76% experienced graft loss and 59% died. Extrapulmonary involvement in all 12 cases of aspergillosis
|
|
Singh, et al., 20123, University of Pittsburgh, Mount Sinai, Wright State, Miami, Mayo Clinic AZ
|
Comprehensive review article on donor-derived fungal
infections from the American Society of Transplantation Infectious Diseases Community of Practice.
“Donors with active invasive mould infections are not considered suitable for organ procurement.”
|
Clinical presentation often associated with
infection at the graft site (fungal arteritis, mycotic aneurysms, anastomotic infections, abscess or fungus ball in the kidney). Invasive aspergillosis associated with high rates of graft loss and mortality.
|
|
Gomez, et al., 20134, University of Pittsburgh
|
Review identifying 23 cases of donor-derived filamentous
fungal infections. 91% in kidney transplant recipients, Aspergillus species in 71%, manifesting as vascular complications, allograft dysfunction, and unexplained febrile illness.
|
Rates of graft loss and mortality were 83% and 17%, respectively. Excellent review of the literature.
|
|
Lopez-Medrano, et al., 201612,
University Hospital, Madrid, and the Spanish, Swiss, and European Infectious Disease Societies
|
Multinational case-control study that included 51 kidney transplant recipients at 19 centers diagnosed with invasive pulmonary aspergillosis in the first 180 days posttransplant between 2000 and 2013
|
>60% all-cause mortality rate, recipient risk
factors included chronic obstructive pulmonary disease, delayed graft function, bacteremia, acute rejection, prolonged hospital stay, lymphopenia, and cytomegalovirus infection
|
|
Zhen, et al., 201711, Beijing, China
|
Case report of both kidney recipients developing mixed fungal infections from the same donor
|
One patient died following nephrectomy
and iliac artery reconstruction, other patient developed urine infection only and survived with functioning graft
|
|
Valerio, et al., 20186, University Hospital, Madrid, Spain
|
Case report of both kidney recipients developing early invasive aspergillus infections from the same donor
|
Both patients underwent early allograft
nephrectomy (day 3) and received liposomal amphotericin B followed by voriconazole and did well
|
|
Siddiqui, et al., 20245, Baskent University, Ankara, Turkey
|
Case report of two kidney and one liver recipients
developing invasive aspergillus from the same pediatric
donor
|
Both kidney recipients underwent nephrectomy
and survived; liver recipient survived without the need for graft explant
|
DISCUSSION
Donor transmitted fungal infections are a potentially fatal complication of organ transplantation [3,4]. Donor-derived Aspergillus infection has rarely been reported, in part because identification of an active Aspergillus infection in a donor is a relative if not absolute contraindication for accepting an organ for transplantation at most centers [5,12]. Table 1 chronicles a literature review of cases of donor derived Aspergillus infection in predominantly kidney transplant recipients. Potential organ donors may be at risk for fungal infections because of the environmental ubiquity of fungi, their prolonged state of immunodepression (or immunosuppression related to prior transplant), breaches in aseptic technique associated with multiple invasive interventions or during organ recovery/preservation, protracted length of stay in the intensive care unit, near-drowning as a cause of death, and transplant tourism [3,4,12]. Donor acceptance criteria have become less restrictive over time, to include the successful use of organs from donors with treated systemic bacterial infections, selected viral and fungal infections, and from donors with increased behavioral risks for infection. However, the potential lethality of an invasive fungal infection in an immunosuppressed transplant recipient following induction immunosuppression remains a barrier to organ recovery and utilization in this setting. Most cases of donor-derived Aspergillus infections have occurred when the infection is unsuspected and undiagnosed in the donor [5-12]. Previous cases of donor-derived invasive aspergillosis following kidney transplantation have reported allograft nephrectomy as part of the management algorithm, which unfortunately may still not prevent mortality (Table 1). The availability of more effective and less nephrotoxic anti-fungal agents has permitted consideration for the selective use of organs from donors that are being treated for known fungal infections [3,4]. However, the benefit to risk ratio of such an endeavor is largely unknown. The imidazole derivatives, especially voriconazole and posaconazole, have fungistatic activity against invasive molds such as Aspergillus and are not directly nephrotoxic. However, these agents are primarily metabolized by the cytochrome P450 enzyme pathway and impair metabolism of medications such as tacrolimus. Because tacrolimus has a narrow therapeutic window with significant interindividual and intraindividual differences in pharmacokinetic parameters, it is imperative that tacrolimus dosing and levels be monitored carefully to avoid toxicity and adverse effects. Although amphotericin B is fungicidal and remains an option for treating invasive aspergillosis, it is nephrotoxic and therefore we elected to use the less nephrotoxic imidazole derivatives for prophylaxis (rather than treatment) in these cases. Personalized care according to donor and recipient risk profiles, heightened clinical vigilance, close monitoring in collaboration with Infectious Disease, Nephrology, and Pharmacy specialists, timing and duration of anti-fungal prophylaxis, and the challenge of finding the appropriate level of immunosuppression to prevent both acute rejection or donor-transmitted opportunistic infections are keys to success when expanding donor acceptance criteria beyond generally accepted infectious disease thresholds.
CONFLICT OF INTERESTS/DISCLOSURE
The authors do not have any conflicts of interest to disclose pursuant to this study. This study was approved by the Atrium Health Wake Forest Baptist Institutional Review Board under the auspices of the Cortex Study, Protocol Number 00084139.
AUTHOR CONTRIBUTIONS
Concept/design: AMG, RCM, RJS
Data analysis/interpretation/collection: JEK, SAK, WD, CJW, ACF, RJS
Drafting article: AMG, RCM, RJS Critical revision and approval of article: AMG, RCM, JEK, SAK, WD, CJW, ACF, RJS
Citation
Stratta RJ, Webb CJ, Jay CL, McCracken E, Garner M et al, (2025) Improved Outcomes Following Simultaneous Pancreas-Kidney Transplantation in the Second Decade of the New Millennium. J Nephrol Kidney Dis 6(3): 5.