Late-Onset Anaphylactic Reaction Induced by Patent Blue V During Sentinel Lymph Node Biopsy: A Rare Case Report and Brief Literature Review
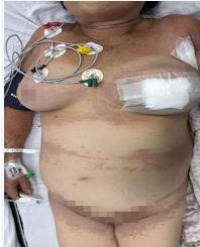
Introduction: Patent Blue V is widely used for sentinel lymph node identification in breast cancer surgery but may cause severe adverse reactions such as anaphylaxis. Although rare, it represents a potentially life-threatening event requiring prompt recognition and management. Methods: A narrative literature review was conducted using PubMed, SciELO, and DOAJ databases, including articles and case reports published between 2005 and 2024 addressing allergic reactions to Patent Blue V. Case Report: A 70-year-old female patient with luminal A invasive carcinoma underwent a segmental mastectomy and sentinel lymph node biopsy (SLNB) after subdermal injection of 2 mL of 2.5% Patent Blue V. In the immediate postoperative period, she developed bluish urticaria, lip edema, and dyspnea without hemodynamic instability. The event was classified as grade II anaphylaxis (Ring and Messmer) and treated with intravenous epinephrine, diphenhydramine, and inhaled salbutamol, with complete resolution within a few hours. Discussion: Anaphylaxis induced by Patent Blue V may occur through immunological mechanisms (IgE-dependent or independent) or direct mast cell activation. Diagnosis is clinical, and prophylaxis remains unvalidated. Systematic reviews suggest a lower risk when using intradermal administration and dye volumes below 2 mL.
Gabriela Conceição Gomes1*, Talita Giuzio¹, Amanda Vicentin Maruya¹, Carlos Alberto Menossi¹, Alice Cunha Lee² and Giovanna Da Conceição Gomes³
