Review Article | Volume 5 - Issue 1 | Article DOI :
Download PDF
Miriam Guest*
Senior Principal Scientific Advisor, Microbial Solutions, Charles River Laboratories, USA
Corresponding Author:
Miriam Guest, Senior Principal Scientific Advisor, Microbial Solutions, Charles River Laboratories, USA
Keywords
Sterility testing; Complex parenterals; Advanced drug delivery systems; Quality control; Pharmaceutical microbiology;
Sterility assurance
Abstract
Advancements in pharmaceutical development have led to increased diversity in drug and biological products. From simple aqueous formulations to advanced drug delivery systems, including sustained release depots, nanospheres and microsphere suspensions, as well as the rise of cell and gene therapy products. With this progress, increased complexity in the manufacture, testing and quality control release of these products has been observed. For injectable products, sterility testing at release is a mandatory step in the product release process. While there are limitations in traditional sterility testing, the final sterility test provides additional sterility assurance for the safety of the product being delivered to the patient. Traditional sterility tests are determined by the visual inspection of nutrient media to assess for the presence of microbial proliferation; product formulations can interfere with visual inspection and ultimately will always lead to a subjective result. Typically, following the initial incubation period, samples of the incubated broth are transferred to fresh nutrient media, and a further incubation step is applied prior to visual inspection.
By Adopting Adenosine Triphosphate (ATP) bioluminescence as the detection platform, the subjective assessment can be avoided, with an unequivocal result determining the presence or absence of microbial proliferation. This technique also avoids the subsequent incubation step on sampling from the sterility test containers.
All living organisms contain the ATP and the enzyme Adenylate Kinase (AK) as part of their biochemical processes, during the ATP Bioluminescence test, an enzyme cascade amplifies the presence of microbial ATP and detect the presence of contamination in the test sample
Background
For the purposes of this review, the phrase “complex parenterals” refers to formulations that are not aqueous formulations. Complex refers to formulations which typically render the nutrient media used in sterility testing turbid, making visual determination of the presence or absence or microbial growth challenging. This encompassing a broad spectrum of products and therapeutic modalities,including advance drugs delivery systems, cellular therapies, biologics, nanosuspensions, vaccines, and products containing challenging adjuvants.Sterility testing a critical release criterion for injectable products, classical microbiological methods are utilised [1,2],whereby samples undergo either membrane filtration, rinsing and the addition of nutrient media or direct inoculation into nutrient media,followed by a 14-day incubation period.Visual inspection of the media is then performed, where the test article is inspected for the presence of microbial growth (e.g., turbidity, pellicle formation, precipitation,flocculent growth or other differences between the test media and the un-inoculated media).Whilst training of personnel executing the sterility test is performed, the results can only ever be interpreted in a subjective manner.
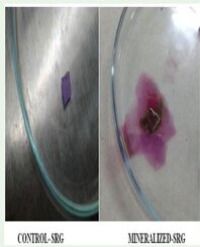
The image (Figure 1) highlights the challenge of visual inspection for a complex formulation and is perhaps one of the worst-case challenges in terms of sample matrix.

Figure 1 Example of Sample Matrix which renders sterility test media turbid.
During traditional sterility testing, a sub-culture step is incorporated at the end of the incubation period, where samples are transferred to fresh broth. During that transfer step, it is possible that product residue could be transferred over to the fresh broth and again make visual inspection for the presence of growth challenging.
The application of ATP bioluminescence requires no additional sub-culture step. The amplified ATP reaction is not impacted by visual artefacts and can provide an unequivocal result of the presence of absence of microbial contamination.
Challenges with Traditional Sterility Testing
Complex Parenterals Membrane filtration is the preferred approach to sterility testing [1,2], which has some benefits over direct inoculation; product residues are removed during the filtration process, whilst potential contamination is captured on the filter membrane, meaning there is less likely to be product related interference impacting microbial proliferation. The nature of the test article should be understood prior to commencing into method development. In the case of complex parenterals, direct inoculation methods are often applied, where samples of the drug product, are placed directly into nutrient media prior to incubation.
During the 14-day incubation period, the broth is visually inspected to assess for the presence of turbidity, pellicle formation, precipitation, flocculant growth and other differences between the test broth and the un-inoculated media. The presence of the drug product itself can cause additional challenges (when compared to simple aqueous formulations undergoing membrane filtrations). The compendia state, that if visual determination cannot be readily assessed, sub-cultures into fresh nutrient media should be conducted. It is prudent to perform both the test set up and the sub-culture steps in a dedicated isolator system, this reduces the risk of cross contamination occurring during the testing itself.
Should growth be suspected, additional sub-culture steps may be applied to fresh nutrient broth and onto plated media. It is recommended that the sub-cultured media is incubated for as long as it took to observe the change in the original test method. Again, this is challenging in products that render the nutrient media turbid, as it may not be obvious when visual growth was originally observed. Sub-culturing is not always reliable [3], and the heavy reliance on these techniques can add additional risk to patient safety.
Sterility by ATP Bioluminescence
The determination of microbial growth by ATP Bioluminescence is a well-established technique, bioluminescence can be observed in nature in glow-worms, marine organisms, and fireflies. The reaction was first described in 1947 [4] and subsequent publications have highlighted the applicability of ATP bioluminescence for the determination of microbial growth [5,6]. All living cells contain ATP and it is a key marker for viable life. The use of ATP bioluminescence in the microbiology laboratory is reliant on the use of a luminometer to determine the light generated and thus, the presence of viable microorganisms in a test article. The technique has been applied for sterility testing, within the pharmaceutical industry with increasing adoption in recent years [7].
The ATP Bioluminescence method amplifies the ATP, and is, therefore, not constrained by the finite amount of metabolic ATP available. Instruments provide automated addition of enzymes, extractants, and buffers, with a luminometer reading the levels of ATP. The amplification technology catalyses and compounds the production of an additional amount of ATP, enabling it to be readily detected. The reaction is essentially linear in nature, a 25-minute reaction time can amplify the amount of ATP by 1000 times more than was originally present. This enables a shortened incubation time, when compared to traditional sterility test methods. Results are reported in Relative Light Units (RLU) and compared to untested broth as a calibrator. Levels of RLU to determine a positive result are established during the method validation process; typically, a three fold increase over the broth calibrator is applied (the broth calibrators will generate low levels of background ATP).
Samples from the broth containers are extracted and transferred in duplicates to cuvettes for testing. Inadvertent cross-contamination at the end of the incubation period will not result in a false positive result, as no additional incubation period takes place, there would be insufficient ATP present to generate an RLU value significantly higher than the broth calibrator.
An overview of traditional sterility testing of complex parenterals is provided in Figure 2.

Figure 2 Traditional Sterility Testing of Complex Parenterals.
Sterility testing involves several manual steps that require technician intervention, each of which increases the risk of cross-contamination during testing. In the event of a sterility test failure or even a suspect positive sterility test result, a complex deviation process ensues.
Sterility test investigations are complex, and often involve a multi disciplinary team. During investigations of suspect-positive sterility test units, production of the drug product may be halted to ensure that root causes are identified and addressed before manufacturing resumes.This can lead to drug shortages and extended downtimes. As with all microbial data deviations, investigations can be prolonged due to the waiting periods required for incubation.
PDA Technical Report 88 [8] provides an overview of the steps required to investigate microbial data deviations. Such deviations include a laboratory review and if no laboratory error is identified, a manufacturing investigation takes place. Some companies may prefer to initiate the manufacturing investigation in parallel, prior to the conclusion of the laboratory investigation.
During the laboratory investigation, a team will consider multiple aspects of the testing:
Equipment:
• Sterility test isolator or laminar flow hood
• Glove integrity testing
• Environmental monitoring data
• Cleanroom certification
• Room conditions acceptable
• Sanitisation and decontamination
Materials:
• Media, QC checks, expiry, storage, integrity
• Could undissolved media cause ambiguous results?
• Could product interference cause ambiguous results?
• Materials transfer
• Glassware management (cleaning & sterilization)
Test Procedure:
• Unusual events documented during testing?
• Complexity of the test
• Sufficient environmental monitoring to detect low levels of background contamination?
• Method suitability conducted appropriately.
Personnel:
• Personnel setting up equipment for the test?
• Personnel performing the test?
• Personnel performing subcultures (if used)?
• Personnel interpreting the test results?
This detailed investigation will require appropriate quality oversight and review.
Determining the presence of absence of microbial growth,by utilizing ATP Bioluminescence can significantly simplify both the testing and investigation process.By reducing manual steps prior to subsequent incubation steps,the risk of extraneous cross-contamination during testing is minimized,thereby reducing the risk of false positives, and leading to simplified investigations.Refer to Figure 3 for an overview of sterility testing by ATP bioluminescence.

Figure 3 Overview of Sterility Testing with ATP Bioluminescence.
Investigational Tool
The ATP Bioluminescence test could be used as an investigative tool, where indeterminate results may stop production until fully investigated. In such cases, the investigation should consider a holistic data set, with a result from an ATP readout being a powerful tool.
Before adopting any change in the QC Microbiology laboratory, a risk assessment should be conducted to understand the proposed future state. In the circumstance of sterility testing, assuming the current method is demonstrated as being suitable for use, as per USP, additional testing could be performed to provide assurance that the ATP bioluminescence response is not impacted by the test article.
In such circumstances, it is anticipated that the sample has undergone a 14-day incubation period, during which period, microbial growth would have generated large quantities of microbial cells.
Adopting an alternative method for investigation purposes may be justified with sound scientific rationale, during investigations a holistic data set should be reviewed. By utilising a non-subjective arbiter of the presence of microbial proliferation, investigations can be expedited. Faster investigations can have broader benefits, by accelerating understanding of the issues faced, the root cause can be more easily identified (by reducing the time to result, the likelihood of detecting the root cause is increased), furthermore, an unequivocal answer on the presence of microbial growth can reduce the manufacturing facility downtime, which may be incurred if a suspect-positive sterility test is observed.
Approach to Method Validation
Should an end user wish to routinely adopt the ATP bioluminescence test, the validation of an alternative microbial method is required. Considerations for the regions where the product sold should be made, but broadly speaking, the application of USP , Ph Eur 5.1.6 and PDA TR 33 should be considered.
Alternative method validation typically directly compares the classical method with the proposed alternative method with the aim to demonstrate non-inferiority of the new method. For qualitative tests, such as sterility, Specificity, limit of detection, ruggedness and robustness should be addressed. It is also important to demonstrate that the test is fit for the intended use; in the case of drug product sample matrices, demonstrating that the test sample matrix does not adversely impact the final result.
Conclusions
Advances in the production of medicinal products has impacts across the manufacturing, testing, and release phases. For advanced drug delivery mechanisms, additional layers of complexity can be observed in the traditional sterility test. By adopting alternative microbiological test methods, such as ATP bioluminescence for complex parenteral formulation, some of these challenges can be overcome, removing subjectivity in test interpretation, and reducing manual activities during the test. Reducing risks of cross contamination during testing can ultimately simplify investigations and can result in reduction of manufacturing downtime and expedite the process of releasing drug products to patients.
References
1. United States Pharmacopeia Sterility Tests.
2.European Pharmacopeia 2.6.1 Sterility Tests.
3.Guest M, Pickard B, Smith B, Drinkwater S. The use of amplified ATP bioluminescence for rapid sterility testing of drug product formulations. PDA J Pharm Sci Technol. 2023; 77: 402-411.
4.McElroy WD. The energy source for bioluminescence in an isolated system. Proc Natl Acad Sci U S A. 1947; 33: 342-345.
5.Stanley PE. A review of bioluminescent ATP techniques in rapid microbiology. J Biolumin Chemilumin. 1989; 4: 375-380.
6.Rapid microbiological methods in the pharmaceutical industry. Martin C Easter Editor. CRC Press. 2003: 288.
7.Deutschmann S, Paul M, Claassen-Willemse M, van den Berg J, IJzerman-Boon P, Grunert da Fonseca V, et al. Rapid sterility test systems in the pharmaceutical industry: Applying a structured approach to their evaluation, validation and global implementation. PDA J Pharm Sci Technol. 2023; 77: 211-235.
8.PDA Technical Report No. 88. Microbial Data Deviations.
9.United States Pharmacopeia Validation of Alternative Microbiology Methods.
10. European Pharmacopoeia 5.1.6. Alternative Methods for Control of Microbiological Quality.
11. PDA Technical Report No. 33 (Revised 2013) Evaluation, Validation and Implementation of Alternative Rapid Microbiological Methods.
Citation
Guest M. (2024) Approaches to Method Development, Suitability and Validation for Alternative Sterility Test Methods for Complex Parenter als. Ann Appl Microbiol Biotechnol J 5: 4.