INTRODUCTION
Cushing Syndrome (CS) is an endocrine disease characterized by hypercortisolemia, with an annual incidence of about 2-3/106, of which Cushing’s Disease (CD) accounts for about 70% of the etiology [1]. CS not only directly affects the metabolism and electrolyte balance of sugar, fat, protein and other substances, but also affects the function of multiple system organs in the body. The hypercoagulable state in CS significantly increases the risk of venous thromboembolic events (VTEs), and evidence shows that CS patients have about a 10-fold higher VTE risk compared to the general population [2,3]. This article summarizes five recent cases of Cushing’s syndrome complicated by pulmonary embolism in our hospital and reviews the literature.
CASE 1
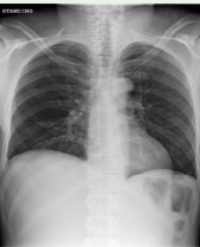
A 36 year old female was admitted to the hospital on November 9, 2021, with complaints of a moon face, weight gain, and an increased abdominal circumference over 7 months. Physical examination: height 162cm, weight 72kg, waist circumference 101cm, BMI 27.43kg/m2, full moon face, central obesity, thin skin all over the body, bloody, scattered acne on the chest and back, and purple lines on the abdominal skin. Functional tests including high-dose dexamethasone suppression test (DST) and desmopressin (DDAVP) stimulation test supported Cushing’s disease. Enhanced pituitary magnetic resonance (MRI) showed abnormal signals in the left and lower parts of the pituitary gland, nature: pituitary tumor? Rathke cysts are not excluded. On November 23, 2021, endoscopic transnasal mid-skull base resection was performed under general anesthesia. Postoperative pathological diagnosis: pituitary adenoma (corticotropin adenoma), immunohistochemistry: tumor CK8/18 (+), syn (+), ACTH (+), TSH (-), GH (-), LH The positive rate of (-), PRL (-), FSH (-), and Ki-67 was about 3%, and tumor reticulum fibers were reduced. Postoperative re-examination of bilateral lower extremity venous color Doppler ultrasonography: partial intermuscular venous thrombosis of bilateral calf. On the fourth postoperative day (November 27th, 2021), the patient suffered sudden dyspnea. Coagulation routine: PT 10.2s, APTT 22.0s, fibrinogen 7.28g/L, fibrin and fibrinogen degradation products 4.7mg/L, D-dimer 1.65mg/L. Blood gas analysis showed no obvious abnormality. Emergency pulmonary artery three-dimensional reconstruction enhanced computed tomography (Computed tomography angiography, CTA): pulmonary artery trunk, left and right pulmonary artery trunk and proximal large branches no clear filling defect. Inflammation and partial atelectasis were scattered in both lungs. After multidisciplinary team consultation, pulmonary embolism could not be ruled out, and meropenem was administered for infection control, and low molecular weight heparin (enoxaparin sodium injection) 0.4 mL subcutaneously was given for 6 days, followed by rivaroxaban 15 mg twice daily for anticoagulation. Repeated chest CT showed that the bilateral lung infection was better than before. D-dimer 0.92mmol/L. She was smoothly transferred to a local hospital for further treatment.
CASE 2
A female, 31 years old, admitted to the hospital on April 6, 2022, for a 1-year history of weight gain, facial puffiness, and 6 months of acne. Specialist physical examination: Height 160.5cm, weight 54kg, waist circumference 86cm, BMI 20.9kg/cm2, full moon face, meager skin, facial acne, bloody face, increased vellus hair on both upper arms, slightly distended abdomen, no purple streaks. The pituitary-target gland function evaluation was perfected after admission. Functional tests such as DST) and DDAVP stimulation test supported Cushing’s disease. Enhanced MRI of the pituitary: the left side of the pituitary sees a first grade T1, slightly shorter T2 signal nodule with a size of about 0.3 × 0.3 cm, which appears to be slightly enhanced after enhancement, and has a clear boundary. The pituitary stalk was not offset. Suspicious nodules on the left side of the pituitary. On April 22, 2022, under general anesthesia, endoscopic transnasal mass resection of the skull base and sellar area was performed. Postoperative pathological diagnosis: pituitary adenoma corticotropin adenoma), immunohistochemistry: ACTH (+), GH (-), TSH (-), PRL (-), FSH (-), LH (-), Syn (+), CK8/18 (+), PIT-1 (-), SF (-), Ki67 positive rate was about 4%, tumor reticulum fibers decreased. Postoperative re-examination of lower extremity venous ultrasonography showed reflux of the right common femoral vein and great saphenous vein. The venous blood flow of the right lower extremity was slow. DIC routine examination: PT 10.2s, APTT 32.7s, fibrinogen 4.64g/L, fibrin and fibrinogen degradation products 5.8mg/L, D-dimer 2.12mg/L. On the 6th postoperative day (April 28, 2022), pulmonary artery CTA: a small filling defect was seen in the pulmonary artery branch of the basal segment of the right lower lobe, mostly due to pulmonary embolism. Blood gas analysis showed no obvious abnormality. The patient received treatment with subcutaneous injection of 0.4ml qd of low molecular heparin (enoxaparin sodium injection), re-examination of DIC routine: PT 10.8s, APTT 25.4s, fibrinogen 4.06g/L, fibrin and fibrinogen degradation products <2.5mg /L, D-dimer 0.60mg/L, no obvious abnormality in blood routine, in stable condition and was discharged from the hospital on Rivaroxaban 10 mg once daily for anticoagulation. About 3 months after the operation (July 18, 2022), no obvious abnormality was found in the re-examination of pulmonary artery computed tomography angiography (CTA).
CASE 3
An 81-year-old female was admitted on April 16, 2022, with a 20 year history of hypertension, bilateral lower extremity edema, and 6 months of elevated blood glucose. Specialist physical examination: height 166cm, weight 73kg, BMI 26.49kg/m2, low hairline, increased beard on the upper lip, fat accumulation in the neck, back and abdomen, scattered ecchymosis on the right forearm and the back and abdomen of the left hand, no skin purple striations, moist rales could be heard in the right lower lung, arrhythmia, premature beats could be heard, pitting edema of both lower extremities, and heavier left lower extremities. The pituitary-target gland function evaluation was perfected after admission. Functional tests supported Cushing’s disease. Ultrasound of lower extremity blood vessels: venous thrombosis in the left part of the calf. Enhanced pituitary MRI: a weakly enhanced nodule with a size of about 0.9cmx0.9cmx0.6cm was seen on the right side of the pituitary, which was close to the cavernous sinus segment of the right internal carotid artery. Pituitary stalk centered. Cerebrospinal fluid signal shadow can be seen in the sella. After admission, she received insulin to control blood sugar, antihypertensive, anti-infection, correct hypokalemia therapy, and improve cardiac function. On April 27, the D-dimer was detected at 13.68 mg/L, which was significantly higher than before, and PT and APTT were no higher than before. significantly changed. The fluctuation of D-dimer after monitoring was about 12mg/L. Rechecked blood gas analysis PH 7.420, PO2 96mmHg, PCO2 45.1 mmHg, lactate 2.18mmol/L, blood oxygen saturation 98.2%, no obvious abnormality was found. The pulmonary artery CTA was completed on May 4: the pulmonary artery trunk was thickened, with a maximum diameter of about 3.7 cm. Filling defects were seen in the pulmonary artery and branch lumens in the basal segment of the right middle lobe and lower lobe, which were mostly pulmonary embolism. After a multidisciplinary discussion on the patient’s condition and bleeding risk, subcutaneous injection of low molecular heparin (enoxaparin sodium injection) or supercillin (nadroparin calcium injection) 0.4-0.6ml q12h was given, and the dose was gradually reduced to 0.4ml per day. Subcutaneous injection, recheck DIC: PT 10.0s, APTT 28.3s, D-dimer 3.13mg/L, fibrinogen 1.62g/L, fibrin and fibrinogen degradation products 6.8mg/L. The blood platelets were gradually decreased to 91x10^9/L, and the subcutaneous injection of Huangdaparinux 2.5mg/day was adjusted according to the patient’s condition. At the same time, elastic stockings were worn. The patients were encouraged to regularly perform plantar and dorsiflexion exercises, and appropriate bedside activities. After many multidisciplinary team consultations, she was treated with Gamma-knife therapy under local anesthesia on May 17, and transferred to a local hospital for further treatment.
CASE 4
A 49-year-old female was admitted on February 21, 2024, with a 2-year history of hypertension and a suspected pituitary lesion for over 1 month. Height: 161cm, weight: 73kg, BMI: 28.16 kg/m2, waist circumference: 104cm, hip circumference: 102cm, full moon face, buffalo back, appearance of bloodiness, bilateral visual acuity and visual field rough test were normal, both eyes orbital edema, cardiopulmonary abnormalities were not found. The abdomen is swollen, a small amount of purple stripes can be seen on the abdomen skin, and the armpit hair and pubic hair are sparse, and the external genital development is normal adult female. Functional tests supported Cushing’s disease. MRI pituitary high-resolution enhanced scan: First class T1 and slightly shorter T2 signal nodules were seen on the left side of the lower part of the pituitary, about 0.6*0.4*0.4cm. After enhancement, the enhancement was slightly uneven, and the degree of enhancement was lower than that of the surrounding glands. The enhancement was uneven in the rest of the pituitary. The pituitary stalk is slightly shifted to the left. No abnormal signal was found in optic chiasma and bilateral cavernous sinus. On March 7, 2024, endoscopic transsphenoidal Surgery was performed under general anesthesia, cerebrospinal fluid leakage was repaired, and bilateral optic neural tube decompression was performed. Postoperative pathological diagnosis: pituitary neuroendocrine tumor, immunohistochemical results: ACTH (+), FSH (+), TSH (-), PRL (-), GH (small number of cells +), LH (-), Syn (+), CK8&18 (+), Ki-67 (MIB-1) (+, < 1%), T-PIL (+), SF-1 (-), PIL-1 (-), Supporting pituitary adrenocorticotropin cell tumor. Venous ultrasonography of both lower limbs was reviewed after surgery: partial intermuscular veins of both lower legs and left great saphenous vein thrombosis. On the seventh day after surgery (March 14, 2024), the patient developed dyspnea, chest tightness, and shortness of breath. Coagulation routine: PT 10.7s, APTT 29.2s, fibrinogen 4.66g/L, D-dimer 2.03mg/L. Pulmonary artery 3D reconstruction enhanced Computed tomography (CTA) showed multiple nodular and flaky filling defects in the right upper pulmonary artery, right middle artery and some of its distal branches. No exact filling defect was found in the pulmonary trunk and the remaining segmental branches, and the main pulmonary artery was thickened, about 3.5cm at the widest point. The pulmonary venous remains were drained into the left atrium in the form of four main arteries, and no exact filling defect was found in the pulmonary vein, left atrium and left atrial appendage. No abnormality was found in color ultrasound of both upper limbs. According to the advice of the respiratory department, enoxaparin sodium 0.6ml bid was injected subcutaneously. On March 20, she experienced nasal bleeding, so the anticoagulation regimen was adjusted to enoxaparin 0.4ml bid, and on March 22, it was changed to Rivaroxaban 10mg qd. During anticoagulant treatment, the coagulation function and blood gas analysis were dynamically monitored, D-dimer fluctuated between 0.87-2.03mg/l FEU, blood gas analysis showed oxygen saturation fluctuated between 98% and 99%, and coagulation routine examination was performed, PT 11.2s, APTT32.4s, etc. Fibrinogen 3.29g/L, fibrin and fibrinogen degradation products <2.5mg/L, D-dimer 0.32mg/L, the anticoagulation regimen after discharge was rivaroxaban 10mg qd, and the coagulation function was normal after regular outpatient follow-up.
CASE 5
A 54-year-old female was admitted on May 28, 2024, after an intracranial lesion had been detected for over 6 months. Relevant examinations were completed after admission, PTC-8 466nmol/l, ACTH 42.97ng/l, 24hUFC 2066ug/24hr. Sellar MRI: Intrasellar and suprasellar occupying, considering pituitary adenoma. On May 30, 2024, endoscopic transnasal middle skull base resection under general anesthesia was performed successfully. Postoperative pathological diagnosis: pituitary neuroendocrine tumor (adrenocorticotropin adenoma), immunohistochemical results: CK8&18 (+), Syn (+), Ki-67 (+, about 2%), SF-1 (-), T-PIT (+), PIT-1 (-), FSH (-), LH (-), ACTH (+), GH (-), PRL (-), TSH (-). 20 days after surgery, she was admitted to Sichuan People’s Hospital due to “repeated fever for 5+ days, aggravated with fatigue for 1 day”, and pulmonary artery CTA was improved. Imaging revealed filling defects in the anterior pulmonary artery of the upper lobe of the right lung, the main middle artery and the lateral pulmonary artery, the outer basal segment of the lower lobe, and pulmonary artery lumen of the anterior basal segment of the left lower lobe, consistent with pulmonary embolism. The patient’s condition improved after symptomatic treatment such as anti infection, anticoagulation and correction of electrolyte disturbance. After discharge, rivaroxaban was regularly taken orally for anticoagulation. No significant filling defect was found in pulmonary artery CTA after outpatient follow-up review.
DISCUSSION
Cushing’s syndrome (C) can affect multiple organ systems, including the hematologic system. Patients with CD often exhibit a hypercoagulable state, and when surgery, anesthesia, and postoperative bed rest are added, the risk of thrombosis rises substantially. Previous reports suggest that up to 20% of individuals with Cushing’s disease develop postoperative venous thrombosis [1]. Pulmonary thromboembolism (PTE) is closely linked to deep vein thrombosis (DVT), and most research on PTE risk factors and outcomes derives from venous thromboembolism studies. According to one meta-analysis, the odds ratio (OR) of VTEs in CS patients compared with the general population is 17.82 [4]. Likewise, a cohort study of 473 patients with CS (including 360 with Cushing’s disease) found a VTE incidence of 14.6 per 1,000 person-years, involving 12 cases of DVT, 15 cases of PTE, and 6 cases of both [5]. Notably, VTEs can also occur in atypical sites such as the mesenteric or jugular veins. Another retrospective analysis revealed that about one-third of VTEs happened before surgery, and roughly 40% emerged within 60 days postoperatively [6]. Moreover, data indicate a significantly higher rate of postoperative VTEs in ACTH-dependent CS patients undergoing pituitary surgery than in those with nonfunctioning pituitary adenomas (NFPA) treated via transsphenoidal surgery [5]. Similarly, Babic et al., reported that adrenalectomy in CS patients significantly increases VTEs risk compared to adrenalectomy for other conditions [7]. These findings underscore the hypothesis that excessive cortisol drives the heightened thrombotic risk in CS. Indeed, in most (but not all) studies, higher cortisol levels correlate with a greater risk of thrombotic events [4-9]. The formation of venous thrombosis is a complex process, and any factors that can lead to stasis of venous blood flow, vascular endothelial injury and blood hypercoagulability are risk factors for VTEs. Several studies have shown that compared with normal controls, the plasma prothrombin time (PT) of CS patients is basically unchanged [10]. The level of activated partial thromboplastin time (APTT) decreases, which reflects the activation of the endogenous coagulation system in the high cortisol state [10-13], and the decline in APTT is more frequent in CS patients with VTE [2]. To further explore the reasons for the decline in APTT, consider the elevated coagulation factor-5 (F-V), coagulation factor-8 (F-VIII), coagulation factor-9 (F-VII), and coagulation factor-12 (F-XII) and von Willebrand factor (vWF) [11-15]. The most striking changes in Cushing’s syndrome are changes in FVIII and vWF [14,15], with studies showing that elevated FVIII levels are associated with a clinically relevant risk of VTE [2-16], while vWF levels are associated with high cortisol levels The degree of hyperemia was positively correlated[17]. Meanwhile, single nucleotide polymorphisms (SNPs) of the VWF gene promoter (especially haplotypes 1-2659A, -2525G) and shorter dinucleotide GT repeat lengths have been shown to be associated with higher vWF levels in CS [18]. After surgery, the remission of Cushing’s syndrome can return to normal vWF, on the contrary, there is no significant change in vWF of Cushing’s syndrome not relieved [11]. In other studies, antithrombin such as C-1 inhibitor, protein C, protein S, and antithrombin III were elevated compared to normal control [11-17]. The increase may be a reactive protective mechanism of the body against the hypercoagulable state. Compared with controls, CS patients have reported decreased levels of thrombin-activated fibrinolysis inhibitor (TAFI), plasminogen activator inhibitor-1 (PAI-1), and α2-plasmin inhibitor, and thrombolytic time delayed, suggesting that patients with CS have reduced fibrinolysis [11-19]. In terms of platelet function, a small number of studies have shown that increased platelet levels and aggregation in CS patients may be associated with increased biosynthesis of thromboxane A2, which promotes platelet release and activation [20]. In addition, the decline of endothelial function in hypercortisolemia state may also be involved in the occurrence of VTEs in CS patients [21]. The clinical assessment of VTE risk in CS patients is complex, and despite changes in parameters such as hemostasis in CS patients, there is no clear evidence that changes in coagulation parameters can determine the risk of VTE in CS patients. Retrospective analysis of perioperative Cushing syndrome patients with pulmonary embolism suggests that a longer course of disease and unrelieved postoperative hypercortisolemia may be risk factors for perioperative pulmonary embolism in CS patients. Patients with elevated preoperative fibrinogen and decreased postoperative red blood cell count and hemoglobin may be at higher risk of pulmonary embolism [22]. The only study to address a model for risk assessment of VTEs in CS patients came from a retrospective analysis by Zilio et al., who presented the CS-VTE score after multiple regression analysis of data from 176 patients with endogenous CS, which identified the following risk factors and scored. Age ≥ 69 points (2 points), reduced mobility (2 points), acute severe infection (1 point), previous cardiovascular events (1 point), midnight blood cortisol exceeding 3.15-folds the upper limit of normal reference (1 point), Shortened APTT (1 point). CS-VTE < 2 points indicate no risk of VTE, CS-VTE = 2 indicates mild risk (10%), CS-VTE = 3 indicates moderate risk (46%), and CS-VTE ≥ 4 indicates high risk (85%)). Using a score of ≥3 as the standard for predicting VTE, the sensitivity is 85% and the specificity is 95%, however, this model of CS-VTE has not been validated in other studie [2]. Despite the increased morbidity and mortality associated with VTE in CS patients, there is no consensus on the indications and treatment regimens for VTE prophylaxis in CS patients. Whether prophylactic anticoagulation can reduce postoperative VTEs in patients with Cushing’s disease and its safety still lack evidence-based medicine. Perioperative use of aspirin for thromboembolism prophylaxis has also been suggested [23]. However, studies on patients with CS are limited. Most scholars support perioperative thrombosis prevention in CS patients. A retrospective study showed that postoperative anticoagulation with unfractionated heparin at 15000-22500 U/d for no less than 2 weeks followed by warfarin for at least 4 months significantly reduced mortality and vascular complications in CS patients [24]. In another study, Barbot et al compared different thromboprophylaxis regimens in patients with Cushing’s disease undergoing transsphenoidal surgery. Group A was given enoxaparin 4000U/d anticoagulation therapy for 14 days after operation, and group B was given low molecular weight heparin anticoagulation for 30 days after operation, while wearing elastic stockings to encourage early activity. Three thrombotic events occurred in group A within 30 days, no related events in group B, and no bleeding events in either group [25]. There is no consensus on the optimal duration of postoperative prophylaxis, though many advocate continuing anticoagulation for approximately one month after discharge. Some scholars believe that the risk of thrombosis is higher 30-60 days after surgery, and it is recommended to continue prevention until 60 days after surgery, especially after bilateral adrenalectomy [25]. There is also a need for great emphasis on nonpharmacological measures, such as wearing compression stockings, intermittent pneumatic leg compression, and early mobilization. In studies of non-CS patients, wearing compression stockings in combination with prophylactic anticoagulation has been shown to reduce the incidence of VTEs [26,27], and bedridden patients should be encouraged to perform regular plantar and dorsiflexion exercises. In addition to therapeutic measures, clinicians must increase vigilance in diagnosing deep vein thrombosis. Untreated deep vein thrombosis can progress to fatal PTE. Clinical symptoms of deep vein thrombosis should be noted. Emboli in PTE can originate from the superior and inferior vena cava pathways or the right heart cavity, most of which originate from the deep veins of the lower extremities. Cases 1 and 3 in this article suggest partial intermuscular venous thrombosis in the lower extremity, and case 2 only suggests slow venous blood flow. Compared with central lower extremity deep vein thrombosis, its clinical manifestations are insidious, and most patients are often asymptomatic, but it may also be one of the sources of pulmonary thrombosis. In conclusion, patients with CS have a significantly higher risk of VTE, and it is prudent to routinely consider VTE prophylaxis and monitoring when diagnosing and treating CS. This article reports 4 cases of Cushing’s disease. In the process of diagnosis and treatment, pulmonary embolism was detected in time through strict clinical observation and monitoring of hemostatic indicators, which brought some enlightenment for the identification, diagnosis and treatment of CS complicated with pulmonary embolism. However, there is currently no consensus on the assessment of the risk of CS patients complicated with VTE, the assessment of the risk of coagulation, the standard plan and time of prevention or treatment, and further experience accumulation and further research are needed in the future.