Review Article
The Novel Applications of Chitosan Nanoparticles for Photodynamic Therapy Technique
Mohammadreza Saboktakin*
1Department of Nanomedicine, NanoBMat Company, Germany
*Corresponding author: Mohammadreza Saboktakin, NanoBMat
Company, GmbH, Hamburg, Germany, Email:
saboktakin123@gmail.com
Submitted: 13 May 2019; Accepted: 28 May 2019; Published: 30 May 2019
Cite this article: Saboktakin M (2019) Medical Applications of Poly Methyl Methacrylate
Nanocomposites. JSMC Nanotechnol Nanomed 3: 5.
Nanoparticles present great potential in drug delivery
applications, yet there are some issues regarding their stability.
In this study was conducted to define the conditions to stabilize
polysaccharide (chitosan/dextran sulfate, CS/DS) nanoparticles
by a process of freeze-drying, assessing the cryoprotectant
capacity of two sugars (sucrose and glucose). Additionally, it was
also intended to find if the solubilisation of chitosan in different
acids affected nanoparticle preparation and characteristics.
Chitosan is obtained by the N-deacetylation of chitin, a polymer
that can be extracted from various sources (crustacean’s shells,
exoskeletons of certain invertebrates – ladybugs – and the cell
walls of fungi, for example). It is considered the second most
abundant polymer on Earth [1], after cellulose [2]. However,
chitin is not very versatile due to its structure and poor solubility
in many solvents. Chitin plays a role in the protection of certain
animals in nature, being organized in semi-crystalline microfibrils
to provide the said protection [3]. The deacetylated form of chitin
is chitosan, which structure can be seen on Figure 1.
Chitosan (MW ~30-190 kDa) is the polymer obtained when
deacetylation surpasses 50% and is comprised of β (1-4)-links of
D-glucosamine and N-acetyl-D- glucosamine monomers that are
distributed randomly throughout the chain. This is the only
natural polymer exhibiting a cationic character [4]. Due to the
fact that the amine groups are present throughout the structure
of chitosan, the more deacetylated the polymer is, the more susceptible it is to protonation because nitrogen has an unused
pair of electrons that can easily interact with electrophilic groups.
When chitosan undergoes protonation, which occurs at low pH
levels, it acquires a positive charge, thus providing the possibility
to interact with negatively charged groups. This ability has been
widely explored in drug delivery, with the preparation of
nanoparticles by electrostatic interaction, as reported using
carrageenan [5], tripolyphosphate (TPP) or dextran sulfate [6] as
counter ions. Chitosan is not soluble in water. Instead, and
because the amine groups have a logarithmic acidity constant
(pKa) of ~6.5, chitosan dissolves easily in acidic media. The most
usual solvent for its dissolution is 1% (v/v) acetic acid. Due to the
fact that the polymer is easily protonated, it can also be dissolved
in formic and lactic acids and in hydrochloric acid solutions [7].
In a pH of 7 or higher, as the pH is higher than the pKa of amine
groups, chitosan becomes insoluble, rendering its biologically
applications are scarce or non-existent. As mentioned earlier,
chitosan presents adequate characteristics regarding biological
applications [8], including biocompatibility, biodegradability and
low toxicity. Its biodegradability is due to a metabolism by
lysozyme [9]. Moreover, due to its protonation in acidic media, it presents a favourable interaction with mucus [10], which is
particularly relevant in the intestine, where there is a high
amount of mucus. This is actually one of the reasons justifying its
application in specific disease conditions, such as colonic
inflammation. Nevertheless, the description of chitosan
applications in literature is endless, including pulmonary and
oral insulin delivery as well as the delivery of antibiotics and
small genetic fragments [11-13], just to mention some examples.
Finally, chitosan can also be found in a variety of dietary
supplements (Lipoforte® and EasySlim® Blocker, just to name a
few) for weight loss. However, the European Food Safety
Authority (EFSA) considers that there is not enough evidence
that supports this indication. There are other studies that report
its beneficial use on lowering LDL-cholesterol [14,15], a claim
that EFSA found to have enough evidence that supports it.
However, some problems have also been described for
supplements, reporting chitosan interference with certain
treatments, for example, anticoagulation therapy with warfarin
[16], and anti-epilepsy therapy with valproate [17]. Dextran
sulfate was the other polymer used in this experimental work. It
is also a natural polymer, obtained from bacteria of the
Leuconostoc genus. It is comprised of a branched an hydroglucose
backbone with attached sulphur groups, the latter believed to be
17% of the total mass of the polymer. This means that, 2.3 sulphur
groups are present for each glucosyl residue, thus the polymer
being expected to be negatively charged. Its chemical structure
can be seen in Figure 2. Dextran sulfate (MW ~5-500 kDa [18-20]
is also reported as biocompatible and biodegradable [21], thus
theoretically complying with mandatory requisites for biomedical
applications. Studies regarding the use of dextran sulfate as a
component of DDS are scarce, even though it was widely studied
in the eighties for its anti-HIV activity, against HIV-1. Its
mechanism of action, in this context, involves inhibiting the
reverse transcriptase, a viral enzyme responsible for turning the
viral RNA in viral DNA that will, ultimately, be part of the host cell
DNA resulting in the production of viral particles [22].
Additionally, dextran sulfate has also been shown to activate
macrophages by establishing an interaction with the scavenger
receptor present in these cells [23]. Moreover, regarding the
immune system, it is believed that dextran sulfate induces
activation of B lymphocytes even though that mechanism is
mediated by macrophages [24]. Another aspect that is widely
mentioned in studies is an anticoagulant capacity similar to
heparin, even though it wasn’t studied further [25]. Finally,
dextran sulfate is used, nowadays, to induce colitis in mouse
models [26,27]. Approved formulations containing this polymer
include two EMA-approved orphan medicines: i) a medicine for
the prevention of graft rejection and its further damage on
pancreatic transplantation by inhibiting the activation of
complement system and clotting [28] and ii) a medicine to
promote mobilisation of progenitor cells, prior to stem cell
transplantation, through a cytokine released by bone marrow
that can guide these important cells through blood stream [29].
Dextran sulfate is a very interesting polymer with many
characteristics that can be used in drug delivery. However, there is still much work to do and, more importantly, study possible
associations with other polymers to produce nanocarriers with a
potential use in therapeutics [30-34]. Chitosan extracted from
Metacarcinus magister shells using demineralization with 1.5M
and 2M HCl showed the characteristic functional groups of CS.
The CS-CMC nanoparticle were chelated with five different
concentrations of TPP and BaCl2 (0.1%, 0.2%, 0.3%, 0.4% and
0.5%) and embodied with CUR. The SEM analysis indicated the
size of the nanoparticles to below 500 nm which was also
confirmed by AFM. The encapsulation efficiency was absolutely
time dependent. The drug release was found to be best with
higher concentration of chelating agents used and the drug
release was prolonging to 80th minute for all the samples. In vitro
drug release showed antibacterial activity when ethanol and
acetic acid was used as solvent system [35]. A new granule
hydrogel composite of natural biocompatible polymeric chitosan
and gelatin (C-G) have prepared. The gelatin side groups
(hydroxyl and carboxyl) can attach with amine groups of chitosan,
which improve the interlink cross linking in the final
nanocomposites. The diameter of the granules is around 3mm to
6 mm. Furthermore, in order to reinforce the mechanical
properties of granules, for the first time, octa (ammonium
chloride) substituted Polyhedral Oligomeric Silsesquioxane C-G/
POSS-(NH3+ Cl-)8 has been used as a nanofiller in the structure of
this nanocomposite. The effect of C-G/POSS-(NH3+ Cl-)8 on mechanical stability and swelling behavior of hydrogels has been
investigated in three pH of 1.2, 7.4 and 9.5, respectively. Results
demonstrate that the C-G/POSS- (NH3+ Cl-)8 granules in acidic pH
has the maximum swelling percent. The encapsulation efficiency
(EE) values for C-G granules in three pH of 1.2, 7.4 and 9.5 are
obtained 65%, 58% and 53% and for C-G/POSS-(NH3+ Cl-)8
sample are 59%, 52% and 48%, respectively, which conform
with the swelling behavior of these hydrogels. According to the
obtained results from Metronidazole (MTZ) release, the assynthesized
hydrogels may have intrinsic ability in the controlled
release of drugs. The as-prepared hydrogels are characterized via
FT-IR, SEM, TGA and XRD analysis [36]. Chitosan nanoparticles
(CS NPs) exhibit good physicochemical properties as drug
delivery systems. Haliza Katas and et al. [37], have determined
the modulation of preparative parameters on the physical
characteristics and colloidal stability of CS NPs. CS NPs were
fabricated by ionic interaction with dextran sulphate (DS) prior
to determination of their storage stability. The smallest CS NPs of
353 ± 23nm with a surface charge of +56.2 ± 1.5mV were
produced when CS and DS were mixed at pH 4 and with a DS: CS
mass ratio of 0.5 : 1. An entrapment efficiency of 98% was
achieved when BSA/siRNA was loaded into the nanoparticles.
The results also showed that particle size and surface charge of
CS NPs were slightly changed up to 2 weeks when stored at 4°C.
Greater particle size and surface charge were obtained with
increasing the concentration of DS. In conclusion, NPs were
sufficiently stable when kept at 4°C and able to carry and protect
protein.
-
 Figure 2: TEM images of CS NPs. (a) and (b) Unloaded CS NPs at 0.5: 1
and 1: 1, (c) and (d) BSA loaded CS NPs at 0.5: 1 and 1: 1, and (e) and
(f) siRNA loaded CS NPs at 0.5 : 1 and 1 : 1, respectively. All the images
were taken at 60 kX magnification. View Figure
Figure 2: TEM images of CS NPs. (a) and (b) Unloaded CS NPs at 0.5: 1
and 1: 1, (c) and (d) BSA loaded CS NPs at 0.5: 1 and 1: 1, and (e) and
(f) siRNA loaded CS NPs at 0.5 : 1 and 1 : 1, respectively. All the images
were taken at 60 kX magnification. View Figure
Ji Woon Suh and et al. [38], have formulated buccal
mucoadhesive nanoparticles (NPs) using the natural
mucoadhesive polymers. The natural mucoadhesive polymers
chitosan (CS) and dextran sulfate sodium salt (DS) were used
to prepare mucoadhesive NPs using the ionic gelation method.
As the molecular weight of DS decreased, the amount of mucin
and the number of buccal cells adsorbed on DS increased. The
CS/DS NPs ranged from 100 to 200nm in diameter. The adhesive
interactions of CS/DS NPs with mucin were not significantly
different from those of CS/sodium triphosphate pentabasic
(TPP) NPs; however, CS/DS NPs exhibited 5 times greater
mucoadhesive activity to buccal cells compared to control CS/
TPP NPs in ex vivo adhesion tests. These results indicate that the
buccal mucoadhesive properties of NPs can be improved using
natural mucoadhesive polymers. A novel nanoparticle delivery
system has been developed by employing the oppositely charged
polymers chitosan (CS) and dextran sulfate (DS), and a simple
coacervation process. Under the conditions investigated, the
weight ratio of the two polymers is identified as a determining
factor controlling particle size, surface charge, entrapment
efficiency and release characteristics of the nanoparticles
produced. Particles of 223 nm mean diameter were produced
under optimal conditions with a zeta potential of approximately
-32.6 mV. A maximum of 75% anti-angiogenesis peptide
entrapment efficiency was achieved with a CS: DS weight ratio of
0.59:1. The same nanoparticle formulation also showed slow and sustained peptide release over a period of 6 days. In contrast, the
formulation containing a lower ratio of CS: DS (0.5:1), was found
to have reduced entrapment efficiency and more rapid peptide
release characteristics. The results suggest that physicochemical
and release characteristics of the CS-DS nanoparticles can be
modulated by changing ratios of two ionic polymers. The novel
CS–DS nanoparticles prepared by the coacervation process
have potential as a carrier for small peptides [39]. Fwu-Long Mi
[40], have prepared chitosan-based nanoparticles containing
indocyanine green (ICG) for hyperthermia therapy and imaging.
ICG was incorporated into chitosan and poly-gamma-glutamic acid
self assembled nanoparticles using the polyelectrolyte complex
method. The ICG encapsulation efficiency of the nanoparticles
was higher than 95% and the ICG-loaded nanoparticles showed
effective temperature elevation upon NIR laser irradiation
(808 nm, 1.5 W/cm2). The cytotoxicity results from NIR laserinduced
hyperthermia effect showed an enhanced toxicity in
overcoming doxorubicin (Dox) resistance of MCF-7/ADR cells.
Pei-Ru Wei et al. [41], have designed a study for photodynamic
therapy (PDT) using chitosan coated Mg-Al layered double
hydroxide (LDH) nanoparticles as the delivery system. A Food
and Drug Administration (FDA) approved near-infrared (NIR)
fluorescent dye, indocyanine green (ICG) with photoactive
properties was intercalated into amine modified LDH interlayers
by ion-exchange. The efficient positively charged polymer
(chitosan (CS)) coating was achieved by the cross linkage using
surface amine groups modified on the LDH nanoparticle surface
with glutaraldehyde as a spacer. The unique hybridization of
organic-inorganic nanocomposites rendered more effective and
successful photodynamic therapy due to the photosensitizer
stabilization in the interlayer of LDH, which prevents the leaching
and metabolization of the photosensitizer in the physiological
conditions. The results indicated that the polymer coating
and the number of polymer coats have a significant impact on
the photo-toxicity of the nano-composites. The double layer
chitosan coated LDH–NH2–ICG nanoparticles exhibited enhanced
photo therapeutic effect compared with uncoated LDH–NH2–
ICG and single layer chitosan-coated LDH–NH2–ICG due to the
enhanced protection to photosensitizers against photo and
thermal degradations. This new class of organic-inorganic hybrid
nanocomposites can potentially serve as a platform for future
non-invasive cancer diagnosis and therapy. A new system have
examined by chitosan (CS)-dextran sulfate (DS) nanoparticles
coated iron oxide as drug carriers detectable using magnetic
resonance imaging (MRI) technique. The 5- aminosalicylic acid
(5-ASA) was chosen as model drug molecule. CS-DS hydrogels
were formulated by a complex coacervation process under mild
conditions. The influence of process variables, including the
two ionic polymers, on particle size, and hydrogel entrapment
of 5-ASA was studied. The in vitro release of 5-ASA were also
evaluated, and the integrity of 5-ASA in the release fraction
was assessed using sodium dodecyl sulfate-polyacrylamide gel
electrophoresis. The release of 5-ASA from hydrogel was based
on the ion-exchange mechanism. The CS–DS hydrogel developed
based on the modulation of ratio show promise as a system for controlled delivery of drug detectable using magnetic resonance
imaging (MRI) technique. Results of release studies indicate that
super paramagnetic chitosan– dextran sulfate hydrogel offer
a high degree of protection from premature drug release in
simulated upper conditions. These hydrogels deliver most of the
drug load in the colon, an environment rich in bacterial enzymes
that degrade the chitosan–dextran sulfate and allow drug release
to occur at the desired site. Thus, spherical super paramagnetic
hydrogel is a potential system for colon delivery of 5- ASA. Also,
this hydrogel can be detected by magnetic resonance imagining
(MRI) technique [42] (Figure 3).
Antony V. Samrot et al. [43], have used chitosan derived from
crab shell using two concentrations of hydrochloric acid i.e.1.5M
and 2M HCl during demineralisation step. The obtained chitosan
was utilized for synthesis of polymeric nanoparticles using
sodium tripolyphosphate and barium chloride as chelators. The
nanoparticles were encapsulated with hydrophobic curcumin
and analysed for drug delivery in vitro. Also, the synthesized
nanoparticles were characterized by FTIR, SEM and AFM analysis
and subjected for drug encapsulation efficiency, in vitro drug
release kinetics and controlled drug delivery studies in vitro against Pseudomonas aeruginosa. Barium chloride was found to
produce spherical shaped drug loaded (Figure 4), nanoparticles
of size below 500nm.
Multifunctional biodegradable and biocompatible chitosan
nanoparticles loaded with drug as an optical-imaging contrast
agent for cancer imaging and as a photothermal therapy agent
for cancer treatment. To improve the efficacy of PTT, chitosan
nanoparticles (NPs) encapsulating super paramagnetic iron
oxide (Fe3O4), drug, and perfluoropentane (PFP) as synergistic
agents for NIR laser-induced PTT.
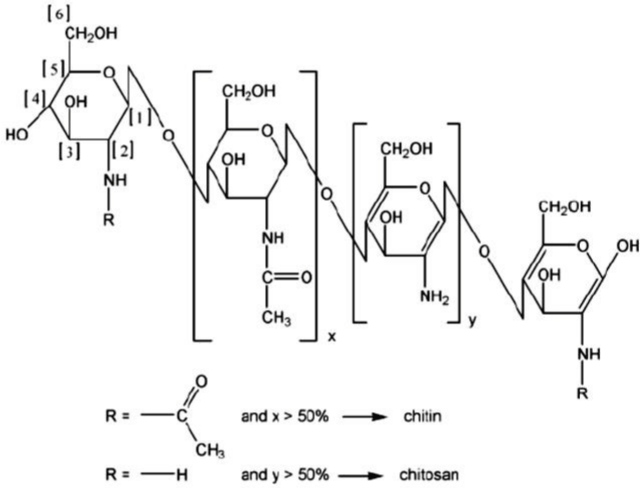 Figure 1: Chemical structure of Chitin and Chitosan. View Figure
Figure 1: Chemical structure of Chitin and Chitosan. View Figure
 Figure 2: TEM images of CS NPs. (a) and (b) Unloaded CS NPs at 0.5: 1
and 1: 1, (c) and (d) BSA loaded CS NPs at 0.5: 1 and 1: 1, and (e) and
(f) siRNA loaded CS NPs at 0.5 : 1 and 1 : 1, respectively. All the images
were taken at 60 kX magnification. View Figure
Figure 2: TEM images of CS NPs. (a) and (b) Unloaded CS NPs at 0.5: 1
and 1: 1, (c) and (d) BSA loaded CS NPs at 0.5: 1 and 1: 1, and (e) and
(f) siRNA loaded CS NPs at 0.5 : 1 and 1 : 1, respectively. All the images
were taken at 60 kX magnification. View Figure
 Figure 3: Morphology of Chitosan-dextran sulfate(CS-DS) hydrogel by
Emission Scanning Microscopy field. View Figure
Figure 3: Morphology of Chitosan-dextran sulfate(CS-DS) hydrogel by
Emission Scanning Microscopy field. View Figure
 Figure 4: Scanning Electron Microscopy analysis of 1.5M demineralized
CS-CMC Nanoparticles and chelated with different concentrations of
BaCl2. View Figure
Figure 4: Scanning Electron Microscopy analysis of 1.5M demineralized
CS-CMC Nanoparticles and chelated with different concentrations of
BaCl2. View Figure