Aspirin has been widely used as a drug for treatment of
inflammation, headache, fever, cardiovascular diseases for
more than a century. Aspirin is well recognized as an effective
anti-platelet drug for secondary prevention in subjects at high
risk of cardiovascular events. However, it is well known that the
side effects of aspirin have limited the using of this drug [1]. The
biochemical mechanism or mode of action of aspirin has been
described previously [2-4]. Also the side effect of aspirin (e.g.
gastrointestinal ulcers) via inhibition of cyclooxygenase (COX)
which is a key enzyme to catalyze prostaglandin formation has
been reported [2]. Recently, aspirin was extended to prevention
and treatment of cardiovascular diseases based on its antithrombotic
action in platelets since inhibition of COX by blocking
thromboxane A2 production which is crucial for blood clotting [5].
High dose aspirin not only lowers the inflammation
mediated pathogenesis of the metabolic syndrome [6], but it
also diminishes hypertriglyceridemia in obese rodents [7], and
patients with type 2 diabetes mellitus [8]. Ahmed et al., reported
that High dose aspirin (120mg/kg), showed maximum decrease
in serum triglyceride level (42.0%), serum total cholesterol level
(19.36%), and serum LDL level (6.18%) [9]. Moreover, Lin HL
et al. reported that low dose aspirin (5 mg/kg) can ameliorate high fat diet induced hyperlipidemia and hyperinsulinemia in
Sprague Dawley rats [10], also suggested that low dose aspirin
may have potential in the prevention of hyperlipidemia induced
lymphocyte adhesion and inflammation. Hua Y et al., also
reported that aspirin (5-20 mg/kg) for 4 weeks can reduce total
cholesterol, triglyceride, LDL and elevate HDL in rabbits which
were given with high fat diet [11].
Hyperlipidemia is a heterogeneous group of disorders
characterized by an excess of lipids in the bloodstream. The
concentrations of lipids, such as triglycerides (TG), cholesterol
(TCH) and low density lipoprotein (LDL) increase, or the level
of high density lipoprotein (HDL) decrease in the blood [12].
Hyperlipidemia is becoming a major health problem in the world
recently even in human and companion animal clinic [13,14].
Chemicals and reagents
CMC-Na (carboxyl methyl cellulose sodium) and simvastatin
was supplied by Tianjin Chemical Reagent Company (Tianjin,
China). Aspirin were supplied by Aladdin Industrial Corporation
(Shanghai China). High fat diet feed (standard rat diet 77.8 %, yolk
power 10 %, lard 10 %, cholesterol 2 %, bile salts 0.2 %) consists
of 41.5 % lipids, 40.2 % carbohydrates, and 18.3 % proteins
(kcal) and standard rat diet consists of 12.3 % lipids, 63.3 %
carbohydrates, and 24.4 % proteins (kcal). The feed was supplied
from KeaoXieli Co., Ltd (Beijing, China). The TG, TC, LDL and HDL
kits were provided by Ningbo Medical System Biotechnology Co.,
Ltd (Ningbo,China). Erba XL-640 analyzer (German) was used to
measure the blood lipid level.
Animals
Wistar male rats with clean grade, aged 7 weeks and
weighing 160–180 g were purchased from the animal breeding
facilities of Lanzhou Army General Hospital (Lanzhou, China).
They were housed in plastic cages of appropriate size (50 × 35
× 20 cm, ten rats per cage) with stainless steel wire cover and
chopped bedding. Light/dark regimen was 12/12 h and living temperature was 22 ± 2°C with relative humidity of 55 ± 10 %.
Rat feed and drinking water were supplied ad libitum. The study
was performed in compliance with the Guidelines for the care
and use of laboratory animals as described in the US National
Institutes of Health and approved by Institutional Animal Care
and Use Committee of Lanzhou Institute of Husbandry and
Pharmaceutical Science of CAAS. Animals were allowed a 2-week
quarantine and acclimation period prior to start of the study.
Drug preparation
Aspirin and simvastatin suspension liquids were prepared in
0.5 % of CMC-Na.
Group and dosing
After their arrival for two weeks, rats were randomized into
two main groups, group I as blank control (n = 10 rats), feed with
basal diet and group II (n = 40 rats) feed with high fat diet (HFD)
for eight weeks. After hyperlipidemias were induced successfully
in rats, the HFD group was averagely divided into four groups
and they were model group and three treatment groups. So it
included three test groups and two control groups as blank and
model groups (Table 1).
The dosage for rats was based on individual weekly body
weights for five weeks. Drugs were administered intragastrically
to each rat. High fat diet (HFD), continued during the experiment
period.
In order to compare Aspirin and simvastatin. For the
comparability of the results in the experiment, The 0.5 % of CMCNa
at the dose of 4ml · kg−1 was as the drug vehicle control and
the dosage of CMC-Na was close to equal in comparison with
Aspirin and simvastatin. Simvastatin (10mg · kg−1) was choosed
as positive control drug to compare with Aspirin (20 mg · kg−1).
At the end of 4th, 6th, 8th, 10th and 13th week after HFD was
used, the rats were fasted for 10-12h and anesthetized with 10%
chloral hydrate. Approximately 1.5ml blood samples were taken
from tail tip for blood lipids examination.
Blood sampling
After fasting for 10-12 h, rats were anesthetized with 10 %
chloral hydrate and blood samples were taken from tail tip for
blood lipids examination. In order to make sure the volume of the
blood sample, the rat tails were immersed in water bath at 45°C
for 5min to make the blood vessel swelling, then approximately
1.5ml blood sample was collected from the rat tail. The serums were got through centrifuge for 15min at the speed of 4000 g at 4°C.
Design of the experiment
First hyperlipidemia model disease is needed to be established
successfully by HFD. At the beginning of the experiment, the rats
were divided into two main groups. Group I feed with standard
diet and Group II feed with HFD. The blood samples were taken
on 4th, 6th and 8th week to examine blood lipids. After the
success of hyperlipidemia animal model, Group II was divided
into four groups, model group and three treatment groups, and
high fat diet was continued during the rest experiment period.
After the administration of drug, blood samples were taken to
measure the change in blood lipids on 10th week and 13th week.
Blood sample was analyzed with the same method.
Statistics
The statistical analyses were carried out using IBM SPSS 19.0
(USA). All data obtained from the experiment are expressed as
mean ± standard deviation (SD). The difference between Group
I and Group II were evaluated by Student’s t-Test. Statistical
differences between the treatments and the control were
evaluated by using one-way ANOVA with Duncan test. P-values
less than 0.05 were considered statistically significant.
Animal disease model
In regard to blood lipid levels, there was no significant
difference between Group I and Group II in week 4 and 6 (data
not shown). So the administration time of HFD was prolonged.
At week 8, there were significant increase of TG, LDL and TC and
decrease of HDL of model group (p < 0.01, seen in Table 2) which
confirmed that the hyperlipidemia animal model was successfully
established by HFD in the experiment.
Body weight
From the results of body weight before drug administration,
blank group had a lower body weight than other groups (P
< 0.01, seen in Table 3), and this indicated that HFD had a
remarkable influence on body weight. There was no significant
difference among other groups except blank group. After drug
administration, the average values of body weight in drug
treatment group were less than model group and there was still
significant difference between blank and model group (P < 0.01). The average body weight values in CMC-Na group were similar
with model group.
Anti-hyperlipidemic effect
Blood samples were taken for lipids examination after the
drugs were given for two weeks, but there was no significant
difference among groups (data not shown). So the time was
extended for five weeks. From Table 4, the difference effects of
drugs on hyperlipidemia appeared after Aspirin was given for five
weeks. Meanwhile, there was no statistical difference between
model and CMC-Na group, which indicated that CMC-Na has no
effect on hyperlipidemia indexes as a vehicle control (Figure 1).
In the blood lipid analysis (seen in Table 4), following five
weeks administration of drugs, TG, TC and LDL were sig-nificantly
decreased in varying degrees in comparison with model group.
These changes meant that there were significant differences
between the treated groups and the model group at the end
of 13th weeks. In regard to TG index (Figure 2), the results in
Aspirin and simvastatin groups were significantly reduced when
compared with model group (p < 0.01). Simvastatin, Aspirin,
reduced significantly levels of LDL (Figure 3) and TC (Figure 4)
(0.01), when compared with model group. HDL in model group
showed no significant difference from blank group at week 13
(Figure 5).
Hyperlipidemia, is a major modifiable risk factor for
atherosclerosis and cardiovascular disease. Increased levels of
LDL are related to the development of atherosclerosis [15,16].
HDL plays an important role in removing cholesterol from tissues
and protecting against cardiovascular disease. Hyperlipidemia
can be the result of an inherited disease in certain breeds of
dogs [13,17]. In pets, hyperlipidemia most often occurs as a
consequence of some disorder, hyperlipidemia even can also
occur spontaneously after a meal of high-fat foods, particularly table scraps [18,19]. In this study, the anti-hyperlipidaemic
effects of Aspirin were investigated in rats with induced
hyperlipidemia by high fat diet (HFD). The blood lipid indexes
were observed during eight weeks. When compared the blood
lipid level between blank and model group at week 8, there was
a significant elevation in the levels of serum LDL, TG and TC, and
decrease of HDL (Table 2). Changed levels of these parameters
in serum are presumptive markers of hyperlipidemia in serum.
LDL is more related to hyperlipidemia and is an important index
in these parameters. On other hand, HDL in this experiment
decreased at week 8 in model group. However, HDL value in
model group showed no significant difference from blank group
at week 13. This apparently unexpected result could be explained
by the light increase of HDL in model group and decrease in blank
group, some studies found that the diet content can effect HDL
level [20], light decrease of HDL in blank group may due to the
feeding long period rich of carbohydrate in standard diet. Some
studies found that the increased plasma apoE of apolipoproteins
in aged hyperlipidemia rats can increase HDL after a long period
[21-25]. The result in this study showed there was significant
change on blood lipid indexes after administrating the drug for
five weeks. The examination of hyperlipidemia indexes after two
weeks of drug administration showed no significant changes
in most groups. So the time was extended to five weeks. The
changes in TC, TG and LDL indexes confirmed that Aspirin had
influence on hyperlipidemia. Previous study on Aspirin showed
that it had effect on TG and TC in Wistar rats with standard diet
[26]. The changes in TC, TG, LDL and HDL are interesting since
they appeared to be most important for hyperlipidemia. This is
very beneficial to the therapy of hyperlipidemia. The result in
(Figure 1), showed that aspirin and simvastatin has similar effect
on hyperlipidemia indexes. From the result in this study, Aspirin
had influence on blood lipid indexes. The changes in TC, TG and
LDL Confirmed that Aspirin had positive influence on regulating
hyperlipidemia. Compared Aspirin with simvastatin and CMC-Na,
it had positive effect on antihyperlipidemia. Aspirin significantly decreased TG, LDL and TC, while simvastatin has similar effect;
this confirmed that Aspirin is benefit on curing hyperlipidemia
based on the results (Table 4 P < 0.01), On the other hand, CMCNa
as a vehicle had no effect on hyperlipidemia, which confirmed
that the influence on hyperlipidemia is due to Aspirin only.
The study on the metabolism of Aspirin showed that Aspirin.
Salicylic acid (SA), from Aspirin metabolism as a final metabolite
excreted in urine was coincided with Salicylic acid from Aspirin
metabolism. The body weight of the rats was increased during
experiment in all groups, which indicated that the rats grew up
healthy during the experiment. These mean the drugs are safety
and no conflict in using on rats.
In addition, Table 4 showed significant differences in the
blood lipid index of the model group compared with the blank group. The action mechanism of simvastatin depends on
inhibition of enzyme 3-hydroxy-3-methyl gluataryl co enzyme A
HMG-CoA reductase inhibitors [27,28 ], which is play a crucial
role on cholesterol synthesis in liver endogenous pathway.
There are many chemical drugs that could ameliorate
hyperlipidemia such as: statins, fibrates, ezetimibe and nicotinic
acid, but most of them are expensive and have undesirable
effect. So there are increasing interest in alternative drug for the
prevention and treatment of hyperlipidemia. Currently available
hyperlipidemic drugs have been associated with a number of side
effects. Therefore, now it’s important to search for drug that is
less toxic, less expensive, which can provide better safety and
efficacy on a long term usage.
In summary, there were significant differences in blood
lipid indexes throughout the experimental period under the
present study conditions. For Wistar rats, the optimal dose for
curing hyperlipidemia by Aspirin was considered to be 20 mg/
kg/day administrating for five weeks, further studies should
be conducted to investigate its prevention effect and the action
mechanism of Aspirin on antihyperlipidemia.
Conceived and designed the experiments: IK NM JYL YJY.
Performed the experiments: IK NM XWL YJY. Analyzed the data:
IK NM JYL XWL. Contributed reagents/materials/analysis tools:
NM IK XWL YJY. Wrote the manuscript: IK NM JYL YJY XWL. All
authors read and approved the final manuscript.
The work was supported by the National Natural Science
Foundation of China (No.31402254), and special project of
Fundamental Scientific Research Professional Fund for Central
Public Welfare Scientific Research Institutes (1610322015013).

 Table 1:The experimental design in the study.
Table 1:The experimental design in the study. 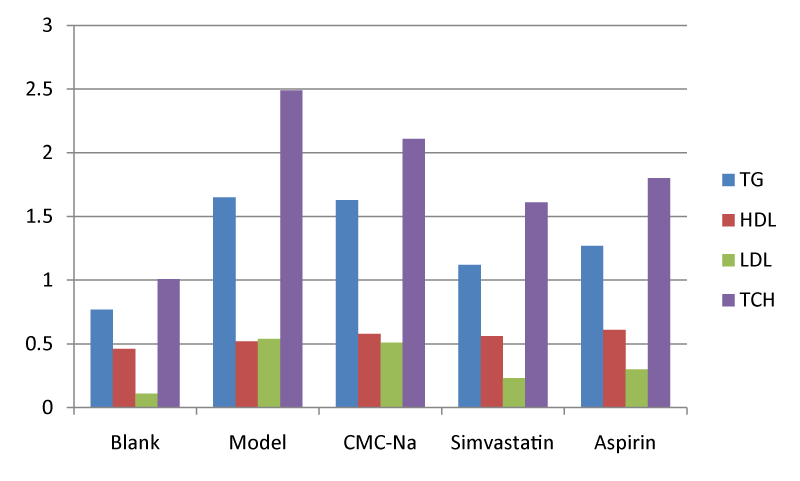 Figure 1: Effects of different drugs on hyperlipemia indexes after
drugs administration for five weeks (n = 10). TG: Triglyceride; HDL:
High density lipoprotein; LDL: Low density lipoprotein; TCH: Total
cholesterol.
Figure 1: Effects of different drugs on hyperlipemia indexes after
drugs administration for five weeks (n = 10). TG: Triglyceride; HDL:
High density lipoprotein; LDL: Low density lipoprotein; TCH: Total
cholesterol. 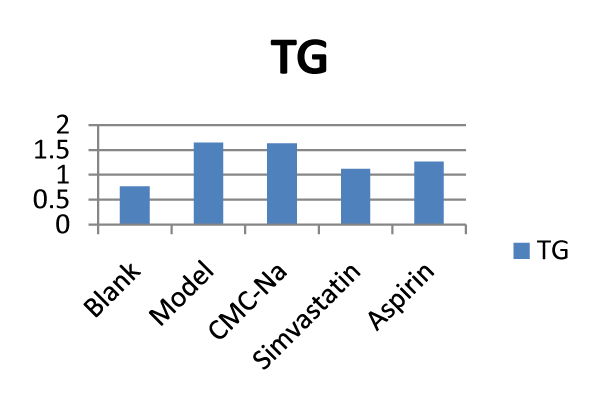 Figure 2: Effects of different drugs on TG: Triglyceride (n = 10).
Figure 2: Effects of different drugs on TG: Triglyceride (n = 10). 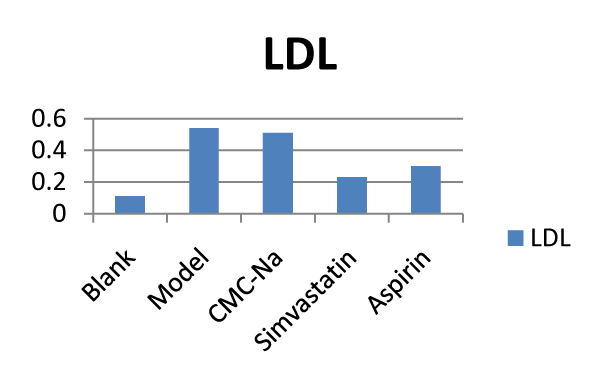 Figure 3: Effects of different drugs on LDL: Low density (n = 10). lipoprotein.
Figure 3: Effects of different drugs on LDL: Low density (n = 10). lipoprotein. 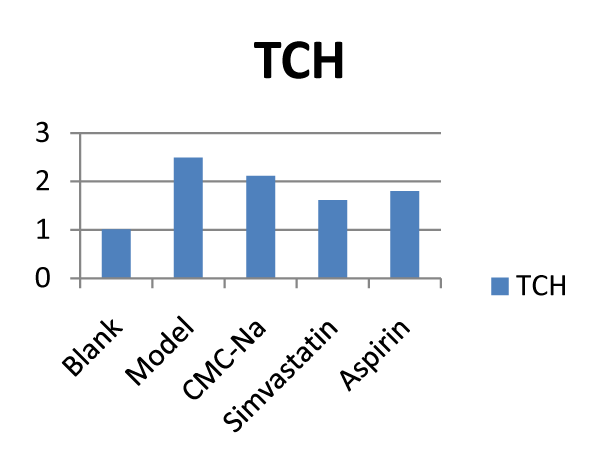 Figure 4: Effects of different drugs on TCH: Total cholesterol (n = 10).
Figure 4: Effects of different drugs on TCH: Total cholesterol (n = 10). 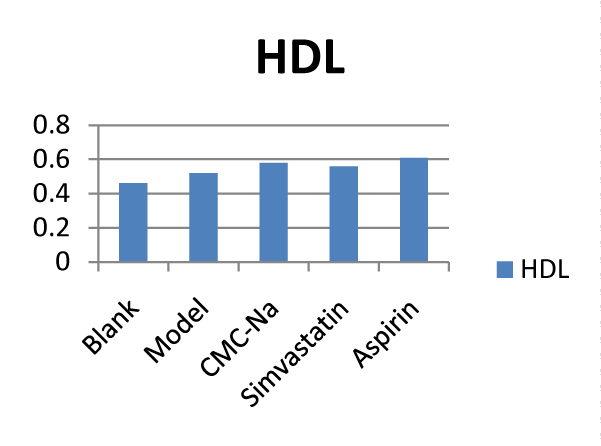 Figure 5: Effects of different drugs on HDL: High density lipoprotein (n = 10).
Figure 5: Effects of different drugs on HDL: High density lipoprotein (n = 10).